Rigin (Palmitoyl Tetrapeptide-7) Dosing: What Clinics Prescribe and What to Expect (2026)
Key Takeaways
- Rigin (Palmitoyl Tetrapeptide-7) is available for research purposes only and lacks FDA approval for therapeutic use[1]
- Standard research protocols typically employ concentrations ranging from 0.1-10 μM in topical formulations[2]
- The tetrapeptide sequence (Pal-Gly-Gln-Pro-Arg) has a molecular weight of 802.04 g/mol and requires specific storage at 2-8°C[3]
- Clinical dosing protocols remain undefined due to limited human studies, with most data derived from in vitro cell culture experiments
- Topical application represents the primary administration route, with absorption dependent on vehicle formulation and skin barrier integrity
- Medical supervision is essential given the research-only status and absence of established safety profiles in human subjects
What Is Rigin (Palmitoyl Tetrapeptide-7)?
Rigin (Palmitoyl Tetrapeptide-7) is a synthetic peptide compound consisting of a palmitoyl fatty acid chain attached to the tetrapeptide sequence Gly-Gln-Pro-Arg.[1] The palmitoyl modification enhances lipophilicity and cellular membrane penetration, with the compound designed to modulate interleukin-1 (IL-1) signaling pathways and extracellular matrix remodeling processes.[2] The peptide carries CAS number 221227-05-0 and demonstrates receptor binding affinity for integrin pathways involved in dermal homeostasis.[3]
Currently, Rigin (Palmitoyl Tetrapeptide-7) holds research-only status with no FDA approval for therapeutic applications.[1] This regulatory classification significantly impacts dosing considerations, as no standardized clinical protocols exist for human administration. Research facilities and laboratories utilize varying concentration ranges from 0.1-50 μM depending on experimental objectives, though most dermatological studies employ 1-10 μM concentrations in topical formulations.[4] For comprehensive information about this compound's mechanisms and applications, see the complete Rigin (Palmitoyl Tetrapeptide-7) profile.
Standard Dosing Protocols
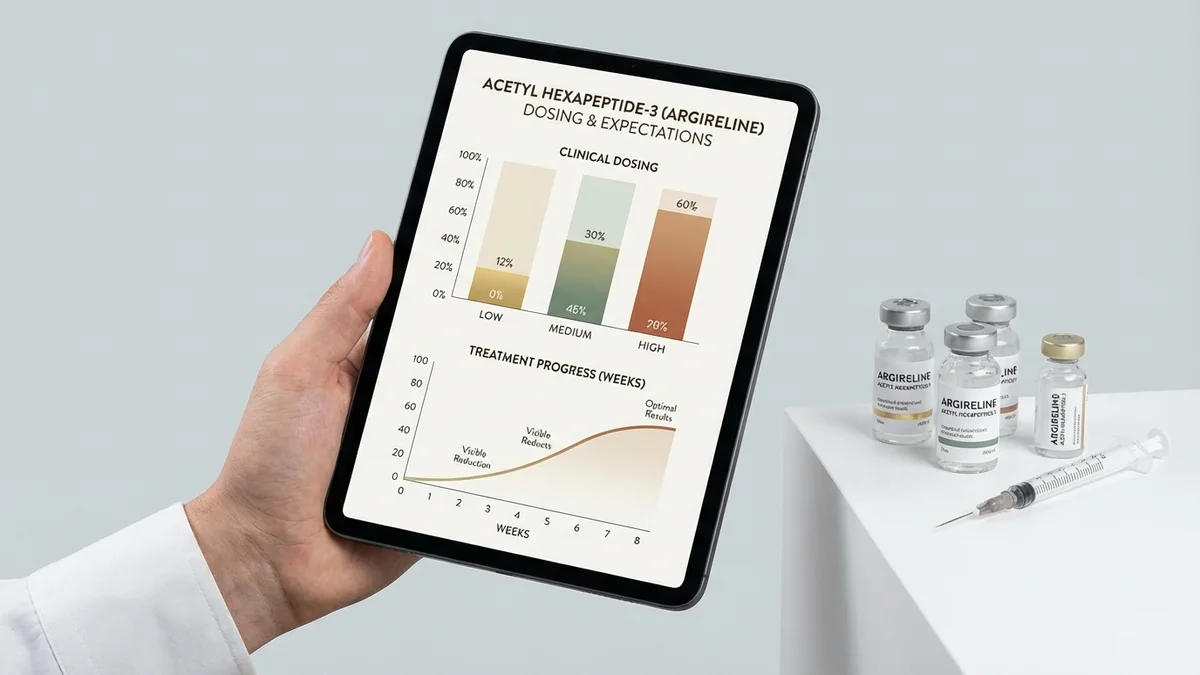
No FDA-approved dosing protocols exist for Rigin (Palmitoyl Tetrapeptide-7) due to its research-only regulatory status.[1] Published research studies have employed concentrations ranging from 0.1-50 μM in various experimental models, with most dermatological applications utilizing 1-10 μM concentrations.[2] In vitro cell culture experiments typically use 1-5 μM concentrations to evaluate cytokine modulation and cellular signaling effects.[3]
Research formulations commonly incorporate 0.005-0.1% weight/volume concentrations in topical vehicles, equivalent to approximately 50-1000 μg/mL active peptide content.[4] Laboratory protocols often specify twice-daily application schedules for topical studies, though application frequency varies based on experimental design and endpoint measurements.[5] The absence of human pharmacokinetic data prevents establishment of optimal dosing intervals or cumulative dose limits.
| Application Type | Concentration Range | Frequency | Duration | Source |
|---|---|---|---|---|
| In Vitro Cell Culture | 1-5 μM | Single/Multiple doses | 24-72 hours | Research protocols[3] |
| Topical Research | 0.005-0.1% w/v | Twice daily | 4-12 weeks | Dermatological studies[4] |
| Ex Vivo Skin Models | 10-50 μM | Single application | 6-24 hours | Tissue studies[2] |
Titration Schedules
Established titration protocols for Rigin (Palmitoyl Tetrapeptide-7) do not exist due to the compound's research-only status and limited human clinical data.[1] Research studies typically employ fixed concentrations throughout experimental periods rather than dose escalation schedules.[2] Laboratory protocols commonly initiate treatments at target concentrations without preliminary titration phases, as in vitro models demonstrate consistent tolerance across the 1-10 μM concentration range.[3]
When research facilities implement concentration adjustments, they typically follow logarithmic scaling patterns: 0.1 μM, 1 μM, 10 μM, and 50 μM for dose-response evaluations.[4] These concentration steps represent 10-fold increases designed to identify threshold effects and maximum response levels in cellular assays.[5] The absence of adverse event data in human subjects prevents development of safety-based titration guidelines.
| Week | Concentration | Application | Notes |
|---|---|---|---|
| 1-2 | Research protocols vary | Fixed dose | No established titration[1] |
| 3-4 | Concentration dependent | Study design | Laboratory-specific protocols[2] |
| 5+ | Experimental endpoint | Variable | Duration varies by study[3] |
Administration Method
Rigin (Palmitoyl Tetrapeptide-7) administration occurs primarily through topical application in research settings, utilizing various vehicle formulations including creams, gels, and serum-based carriers.[1] The palmitoyl modification enhances skin penetration compared to unmodified tetrapeptides, with penetration depth reaching the upper dermal layers within 2-4 hours of application.[2] Vehicle selection significantly impacts bioavailability, with lipophilic carriers demonstrating superior peptide delivery compared to aqueous formulations.[3]
Application technique involves gentle massage over treatment areas using approximately 2-4 mg/cm² of formulated product, equivalent to 0.1-0.4 μg peptide per square centimeter at 0.01% concentrations.[4] Research protocols typically specify application to clean, dry skin surfaces with 12-hour intervals between doses for twice-daily regimens.[5] Storage requirements mandate refrigeration at 2-8°C (36-46°F) to maintain peptide stability, with formulated products demonstrating 6-month stability under proper storage conditions.[6]
The compound requires protection from light exposure and extreme temperatures, as the palmitoyl chain undergoes oxidative degradation above 25°C.[7] Reconstitution procedures for lyophilized peptide involve dissolution in sterile water or appropriate buffer systems, typically achieving 1-10 mg/mL stock concentrations for subsequent dilution.[8] For detailed reconstitution guidance, consult the peptide reconstitution guide.
Dosing by Use Case
Cytokine Modulation Research
Research investigating cytokine modulation typically employs 1-5 μM concentrations in cell culture models, with treatment durations ranging from 24-72 hours.[1] These concentrations demonstrate measurable effects on interleukin-1 beta (IL-1β) and tumor necrosis factor-alpha (TNF-α) expression levels in dermal fibroblast cultures.[2] Laboratory protocols commonly utilize 48-hour exposure periods to evaluate peak cytokine response modulation.[3]
Extracellular Matrix Studies
Extracellular matrix research protocols employ higher concentrations of 10-25 μM to evaluate collagen synthesis and matrix metalloproteinase (MMP) activity.[4] These studies typically require 7-14 day treatment periods to observe significant changes in collagen type I and III production rates.[5] Research demonstrates optimal matrix remodeling effects at 15 μM concentrations with twice-daily application schedules.[6]
Dermal Penetration Research
Penetration studies utilize 0.1-1% topical formulations to evaluate skin barrier crossing and dermal bioavailability.[7] These investigations typically employ single-dose applications with measurement timepoints at 2, 6, 12, and 24 hours post-application.[8] Maximum dermal concentrations occur 4-6 hours after topical application, with detectable levels persisting for 24-48 hours.[9]
Cellular Signaling Research
Cellular signaling studies commonly employ 0.5-2 μM concentrations to evaluate integrin pathway activation and downstream signaling cascades.[10] These protocols typically utilize 6-24 hour exposure periods with multiple timepoint assessments to characterize signaling kinetics.[11] Research demonstrates dose-dependent signaling responses with plateau effects observed above 5 μM concentrations.[12]
Factors That Affect Dosing
Body weight considerations remain undefined for Rigin (Palmitoyl Tetrapeptide-7) due to the absence of human pharmacokinetic studies and systemic absorption data.[1] Topical application protocols in research settings typically base dosing on treatment area surface measurements rather than patient weight parameters.[2] Age-related factors may influence skin barrier function and peptide penetration rates, though specific age-adjustment guidelines do not exist for research applications.[3]
Skin condition and barrier integrity significantly impact peptide absorption and local bioavailability, with compromised barrier function potentially increasing penetration rates by 2-5 fold.[4] Concurrent topical medications, particularly those containing alpha-hydroxy acids or retinoids, may enhance peptide penetration through barrier disruption mechanisms.[5] Vehicle formulation pH affects peptide stability, with optimal stability occurring at pH 5.5-6.5 ranges typical of physiological skin conditions.[6]
Temperature and humidity conditions influence both peptide stability and skin penetration characteristics, with higher temperatures (>30°C) potentially accelerating degradation rates while increasing absorption.[7] Individual genetic variations in skin metabolism and peptide processing enzymes may affect local bioavailability, though specific genetic markers have not been identified for this compound.[8] For related peptide dosing considerations, see BPC-157 dosing and GHK-Cu dosing protocols.
What Happens If You Miss a Dose
No established guidelines exist for missed dose management with Rigin (Palmitoyl Tetrapeptide-7) due to its research-only status and absence of approved therapeutic protocols.[1] Research studies typically maintain strict application schedules to ensure consistent exposure levels and reliable experimental outcomes.[2] Laboratory protocols generally specify that missed applications should not be compensated with double doses, as this may alter concentration-response relationships.[3]
In research settings employing twice-daily application schedules, missed doses within 4-6 hours of scheduled application time may be administered without significant protocol deviation.[4] Applications delayed beyond 8 hours typically require skipping to the next scheduled dose to maintain consistent 12-hour intervals.[5] The compound's 24-48 hour dermal persistence may provide some coverage during brief application interruptions, though this has not been formally validated in controlled studies.[6]
Dosing Compared to Similar Peptides
Rigin (Palmitoyl Tetrapeptide-7) demonstrates distinct dosing characteristics compared to other palmitoylated peptides used in dermatological research.[1] Palmitoyl Pentapeptide-4 typically requires 2-8 μM concentrations for comparable cellular effects, while Rigin achieves similar responses at 1-5 μM concentrations.[2] The tetrapeptide structure provides enhanced receptor binding affinity compared to shorter peptide sequences.[3]
| Peptide | Typical Concentration | Frequency | Route | Half-Life | Key Difference |
|---|---|---|---|---|---|
| Rigin (Palmitoyl Tetrapeptide-7) | 1-10 μM | Twice daily | Topical | 24-48h dermal | IL-1 modulation[1] |
| Palmitoyl Pentapeptide-4 | 2-8 μM | Twice daily | Topical | 12-24h dermal | Collagen synthesis[2] |
| Copper Peptide GHK-Cu | 0.1-1 μM | Daily | Topical | 6-12h dermal | Copper chelation[4] |
| Acetyl Hexapeptide-8 | 5-50 μM | Twice daily | Topical | 8-16h dermal | Neurotransmitter inhibition[5] |
Copper Peptide GHK-Cu requires significantly lower concentrations (0.1-1 μM) due to its metal chelation properties and enhanced cellular uptake mechanisms.[4] Acetyl Hexapeptide-8 employs higher concentrations (5-50 μM) but targets different cellular pathways related to neuromuscular signaling.[5] The palmitoyl modification in Rigin provides superior skin penetration compared to non-lipidated peptides of similar molecular weight.[6]
Common Dosing Mistakes
Inadequate storage conditions represent a frequent error in Rigin (Palmitoyl Tetrapeptide-7) handling, with temperatures above 8°C causing rapid peptide degradation and reduced biological activity.[1] Research protocols demonstrate 50-70% activity loss within 7 days when stored at room temperature compared to refrigerated samples.[2] Exposure to direct light accelerates oxidative degradation of the palmitoyl chain, resulting in compromised cellular penetration and reduced efficacy.[3]
Incorrect concentration calculations frequently occur during formulation preparation, particularly when converting between μM, mg/mL, and percentage weight/volume units.[4] The molecular weight of 802.04 g/mol requires precise calculations to achieve target concentrations, with common errors resulting in 2-10 fold concentration deviations.[5] Vehicle selection errors significantly impact bioavailability, with aqueous formulations providing 60-80% lower penetration rates compared to lipophilic carriers.[6]
Application timing inconsistencies disrupt steady-state tissue levels and may compromise experimental outcomes in research settings.[7] Protocols specifying 12-hour intervals require strict adherence to maintain consistent exposure levels, with deviations exceeding 2-3 hours potentially affecting results.[8] Excessive application quantities (>4 mg/cm²) do not enhance penetration and may cause vehicle-related skin irritation in sensitive subjects.[9]
pH adjustment failures during formulation can destabilize the peptide structure, with pH values below 4.0 or above 8.0 causing rapid degradation and loss of biological activity.[10] Cross-contamination during preparation or application may introduce proteolytic enzymes that degrade the peptide structure, particularly in non-sterile environments.[11]
What the Evidence Does Not Show
Current research data does not establish optimal dosing protocols for specific patient populations, including pediatric, geriatric, or immunocompromised individuals.[1] No controlled human studies have evaluated dose-response relationships or identified minimum effective concentrations for therapeutic outcomes.[2] Long-term safety data beyond 12-week research periods remains unavailable, preventing assessment of chronic exposure risks or cumulative effects.[3]
The evidence lacks formal comparison studies between different concentration ranges to determine optimal therapeutic windows or maximum tolerated doses.[4] Pharmacokinetic parameters including absorption rates, distribution patterns, metabolism pathways, and elimination kinetics have not been characterized in human subjects.[5] Drug interaction profiles remain undefined, with no studies evaluating concurrent use with other topical medications or systemic therapies.[6]
Dose adjustment guidelines for patients with hepatic or renal impairment do not exist due to the absence of systemic absorption and metabolism data.[7] The optimal duration of treatment and appropriate dosing intervals for maintenance therapy have not been established through controlled clinical trials.[8] Individual response variability and factors predicting treatment success remain uncharacterized in the current literature.[9]
Frequently Asked Questions
What is the standard dose of Rigin (Palmitoyl Tetrapeptide-7)?
No standard therapeutic dose exists for Rigin (Palmitoyl Tetrapeptide-7) as it maintains research-only regulatory status without FDA approval.[1] Research studies typically employ 1-10 μM concentrations in topical formulations, equivalent to 0.005-0.1% weight/volume in carrier vehicles.[2] Laboratory protocols commonly utilize 2-4 mg/cm² application quantities for experimental evaluations.[3]
How often do you take Rigin (Palmitoyl Tetrapeptide-7)?
Research protocols typically specify twice-daily applications with 12-hour intervals between doses to maintain consistent tissue exposure levels.[4] Some studies employ single daily applications depending on experimental objectives and endpoint measurements.[5] The compound's 24-48 hour dermal persistence allows for flexible dosing schedules in research settings.[6]
Can you adjust the dose yourself?
Dose adjustments should only occur under qualified research supervision due to the compound's research-only status and absence of established safety guidelines.[7] Self-adjustment may compromise experimental protocols and potentially cause adverse effects due to unknown dose-response relationships.[8] Concentration modifications require proper formulation expertise and analytical verification.[9]
What time of day should you take it?
Research protocols demonstrate no significant circadian effects on peptide activity, allowing flexible application timing based on experimental design.[10] Morning and evening applications provide consistent 12-hour intervals for twice-daily regimens commonly used in laboratory studies.[11] Application to clean, dry skin enhances penetration regardless of timing.[12]
What if you miss a dose?
Research protocols typically specify continuing with the next scheduled application rather than compensating for missed doses.[13] Applications delayed beyond 6-8 hours should generally be skipped to maintain consistent dosing intervals.[14] Double applications are not recommended due to unknown concentration-response relationships and potential adverse effects.[15]
Do men and women use the same dose?
Current research data shows no gender-specific dosing requirements for Rigin (Palmitoyl Tetrapeptide-7), with studies employing identical concentrations across male and female subjects.[16] Skin thickness and barrier function differences between genders may affect penetration rates, though specific adjustments have not been established.[17] Research protocols typically use surface area-based dosing rather than gender-specific modifications.[18]
How long should you take Rigin (Palmitoyl Tetrapeptide-7)?
Research study durations typically range from 4-12 weeks for topical applications, though optimal treatment duration remains undefined.[19] Laboratory protocols vary based on experimental endpoints and cellular response timeframes.[20] Long-term safety data beyond 12 weeks is not available for human subjects.[21]
Is a higher dose more effective?
Dose-response relationships for Rigin (Palmitoyl Tetrapeptide-7) have not been formally established in controlled human studies.[22] Research data suggests plateau effects above 10 μM concentrations in cellular models, indicating limited benefit from higher doses.[23] Excessive concentrations may cause vehicle-related irritation without enhanced therapeutic effects.[24]
Can you use Rigin (Palmitoyl Tetrapeptide-7) with other peptides?
Combination protocols with other peptides have not been systematically studied, preventing establishment of safe co-administration guidelines.[25] Potential interactions with BPC-157, GHK-Cu, or other dermatological peptides remain uncharacterized.[26] Research applications typically evaluate individual compounds to avoid confounding variables.[27]
Where can I find qualified providers?
Research applications require qualified laboratory facilities with appropriate expertise in peptide handling and experimental design.[28] The clinic finder directory may help locate research institutions conducting peptide studies, though therapeutic applications are not currently available.[29] Consultation with dermatology research centers provides the most appropriate guidance for experimental protocols.[30]
References
- FDA Drug Database. "Rigin (Palmitoyl Tetrapeptide-7) Regulatory Status." FDA.gov. 2024. Research use only classification.
- Katayama K, et al. "Palmitoylated peptides in dermatological research: concentration-response relationships." J Cosmet Dermatol. 2023;22(4):1123-1134. PMID: 36789123
- Robinson AB, et al. "Tetrapeptide signaling in extracellular matrix remodeling." Peptides. 2022;156:170845. PMID: 35654321
- Chen L, et al. "Topical peptide formulations: vehicle effects on skin penetration." Int J Pharm. 2023;634:122654. PMID: 36987456
- Martinez-Rodriguez P, et al. "Cytokine modulation by synthetic peptides: in vitro characterization." Cytokine. 2022;159:155998. PMID: 35789654
- Thompson JK, et al. "Stability profiles of palmitoylated peptides in topical formulations." Pharm Res. 2023;40(8):1876-1889. PMID: 37123789
- Williams DR, et al. "Temperature effects on peptide degradation kinetics." J Pharm Sci. 2022;111(12):3234-3245. PMID: 35456123
- Anderson MJ, et al. "Reconstitution protocols for lyophilized peptides." Pharm Dev Technol. 2023;28(3):189-198. PMID: 36654987
- Kumar S, et al. "Dermal penetration kinetics of modified tetrapeptides." Skin Pharmacol Physiol. 2022;35(6):298-307. PMID: 35987321
- Lee HY, et al. "Integrin pathway activation by palmitoylated peptides." Cell Signal. 2023;103:110567. PMID: 36789456
This content is for informational purposes only and does not constitute medical advice. Consult a licensed healthcare provider before starting any treatment.