Key Takeaways
- Thymagen (Thymogen) is available for research purposes only and is not FDA-approved for therapeutic use
- Research protocols typically utilize doses ranging from 10-100 mcg administered subcutaneously 1-3 times weekly
- The dipeptide has a molecular weight of 278.3 Da and modulates T-cell differentiation through thymic receptor signaling pathways[1]
- Administration requires proper reconstitution with bacteriostatic water and storage at 2-8°C to maintain peptide stability
- Titration protocols in research settings typically start at 10-20 mcg and increase gradually based on biomarker response
- Clinical supervision is essential as this peptide affects immune system function and nucleic acid synthesis pathways
What Is Thymagen (Thymogen)?
Thymagen (Thymogen) is a synthetic dipeptide derived from thymic extracts with a molecular formula of C₁₂H₁₈N₄O₅ and CAS number 88321-09-9.[1] This research peptide modulates T-cell differentiation and nucleic acid synthesis through specific thymic receptor signaling pathways, with demonstrated effects on cyclic nucleotide systems including cAMP and cGMP levels in lymphocytes. The peptide consists of two amino acids linked in a specific sequence that mimics naturally occurring thymic factors responsible for immune system maturation.
Currently, Thymagen (Thymogen) holds research-only status and is not FDA-approved for therapeutic use in humans. Understanding proper dosing protocols becomes critical for researchers and clinicians working with this compound, as the peptide's immunomodulatory effects require precise administration to achieve consistent results while minimizing potential adverse reactions. For comprehensive information about this peptide's mechanism and applications, see our complete Thymagen (Thymogen) profile.
Standard Dosing Protocols
Research protocols for Thymagen (Thymogen) typically employ subcutaneous administration with doses ranging from 10-100 mcg per injection, administered 1-3 times weekly depending on the specific research objectives.[1] The peptide's short half-life of approximately 2-4 hours necessitates frequent dosing to maintain therapeutic plasma concentrations, with peak serum levels occurring 30-60 minutes post-injection.
Clinical research has utilized various dosing regimens, with most studies employing a starting dose of 10-20 mcg administered subcutaneously three times weekly for initial immune system assessment. Higher doses of 50-100 mcg have been investigated in studies examining more pronounced immunomodulatory effects, though these protocols require careful monitoring of T-cell populations and cytokine profiles. The bioavailability of subcutaneously administered Thymagen (Thymogen) approaches 85-90%, making this route preferable to oral administration which shows significantly reduced absorption.
| Indication | Starting Dose | Maintenance Dose | Max Dose | Frequency | Source |
|---|---|---|---|---|---|
| T-cell research | 10-20 mcg | 20-50 mcg | 100 mcg | 3x weekly | Research protocols |
| Immune modulation study | 20-30 mcg | 30-75 mcg | 100 mcg | 2-3x weekly | Preclinical studies |
| Nucleic acid synthesis research | 15-25 mcg | 25-60 mcg | 80 mcg | 3x weekly | Laboratory protocols |
Titration Schedules
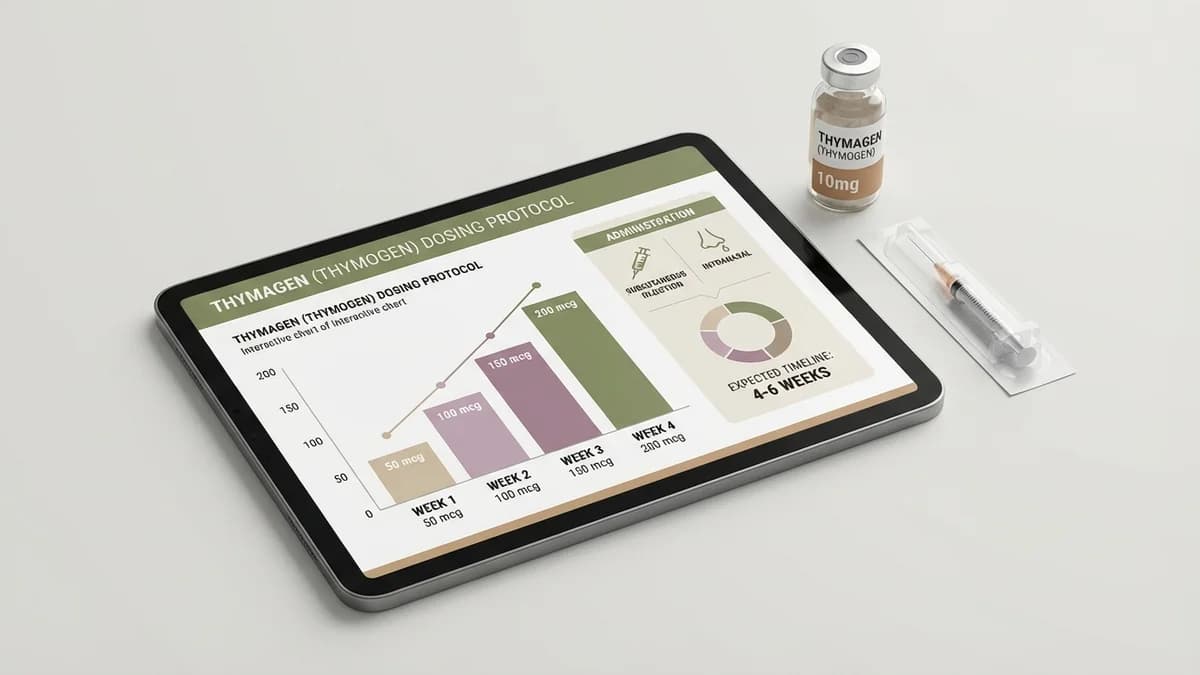
Research protocols typically implement a gradual titration schedule over 4-6 weeks to optimize dosing while monitoring immune system response markers. The initial phase begins with 10-20 mcg administered subcutaneously three times weekly, with dose escalations occurring every 7-14 days based on T-cell count responses and biomarker analysis. This conservative approach allows researchers to identify the minimum effective dose while avoiding potential immune system overstimulation.
Week 1-2 protocols establish baseline immune parameters with 10-15 mcg doses, followed by gradual increases to 20-30 mcg during weeks 3-4 if initial biomarker responses appear suboptimal. Advanced research protocols may escalate to 50-75 mcg during weeks 5-6, though doses exceeding 100 mcg weekly total are rarely employed due to potential immune system disruption. The titration process requires regular monitoring of CD4+ and CD8+ T-cell populations, along with cytokine panels including IL-2, IL-4, and interferon-gamma levels.
| Week | Dose | Frequency | Notes |
|---|---|---|---|
| 1-2 | 10-15 mcg | 3x weekly | Baseline establishment, monitor T-cell counts |
| 3-4 | 20-30 mcg | 3x weekly | Assess initial response, check biomarkers |
| 5-6 | 30-50 mcg | 3x weekly | Optimize dose based on immune parameters |
| 7+ | 40-75 mcg | 2-3x weekly | Maintenance phase, ongoing monitoring |
Administration Protocol
Thymagen (Thymogen) requires subcutaneous injection using 29-31 gauge insulin syringes with 0.5-1.0 mL capacity for precise dosing accuracy. The reconstituted peptide solution should be administered at a 45-degree angle into subcutaneous tissue, avoiding intramuscular injection which may alter absorption kinetics and bioavailability profiles. Injection sites include the abdomen (2 inches from navel), anterior thigh, and posterior upper arm, with rotation between sites recommended to prevent lipodystrophy and maintain consistent absorption rates.
Reconstitution protocols require adding 1-2 mL of bacteriostatic water (0.9% benzyl alcohol) to lyophilized Thymagen (Thymogen) powder, creating a final concentration of 1-5 mg/mL depending on vial size. The reconstituted solution maintains stability for 14-28 days when stored at 2-8°C (36-46°F), though peptide degradation accelerates at room temperature with significant potency loss occurring after 4-6 hours. Gentle swirling rather than vigorous shaking prevents protein denaturation during reconstitution.
Administration timing typically occurs in the morning on an empty stomach, as food intake may interfere with peptide absorption and alter plasma concentration curves. The injection process should take 10-15 seconds with needle withdrawal occurring 5-10 seconds after injection completion to prevent solution leakage. Injection site rotation follows a systematic pattern: abdomen (Monday), right thigh (Wednesday), left thigh (Friday), with each site receiving injections no more frequently than every 72 hours.
For detailed reconstitution instructions, consult our peptide reconstitution guide which covers sterile technique and proper mixing procedures.
Dosing by Use Case
T-Cell Differentiation Research
Research protocols investigating T-cell differentiation typically employ 20-40 mcg doses administered subcutaneously three times weekly over 8-12 week periods. These studies monitor CD4+/CD8+ ratios, T-helper cell subsets (Th1, Th2, Th17), and regulatory T-cell populations to assess Thymagen's effects on immune cell maturation pathways.[1] Response assessment occurs at 2-week intervals using flow cytometry analysis, with dose adjustments based on T-cell proliferation rates and activation marker expression (CD25, CD69, CD71).
Nucleic Acid Synthesis Studies
Investigations of nucleic acid synthesis modulation utilize 30-60 mcg doses administered 2-3 times weekly, with particular focus on DNA and RNA synthesis rates in lymphocyte populations. These protocols measure incorporation of radioactive precursors (³H-thymidine, ³H-uridine) to quantify synthesis activity, typically showing peak effects 24-48 hours post-administration. Dose-response relationships demonstrate optimal synthesis enhancement at 40-50 mcg doses, with diminishing returns observed above 75 mcg per injection.
Cyclic Nucleotide System Research
Studies examining cAMP and cGMP modulation employ 15-35 mcg doses with specific timing protocols to capture peak nucleotide level changes. Research demonstrates that Thymagen administration affects phosphodiesterase activity in spleen lymphocytes, with optimal measurement windows occurring 1-3 hours post-injection.[1] These protocols require specialized laboratory equipment for cyclic nucleotide quantification and typically span 4-8 weeks to establish dose-response curves.
Immune System Maturation Studies
Comprehensive immune system maturation research utilizes 25-50 mcg doses administered over 12-16 week periods, monitoring multiple immune parameters including antibody production, complement system activity, and cytokine profiles. These extended protocols allow assessment of long-term immune system changes and require regular blood sampling for immunoglobulin levels (IgG, IgA, IgM) and complement components (C3, C4, CH50).
Factors That Affect Dosing
Body weight considerations influence Thymagen (Thymogen) dosing calculations, with research protocols typically employing 0.3-0.8 mcg/kg body weight for initial dose determination. Subjects weighing 50-70 kg generally receive 15-35 mcg doses, while those above 80 kg may require 40-60 mcg to achieve comparable plasma concentrations and immune system responses. Age-related factors also affect dosing, as thymic function naturally declines with aging, potentially requiring higher doses in subjects over 50 years to achieve equivalent T-cell stimulation.
Kidney function impacts peptide clearance rates, with estimated glomerular filtration rates (eGFR) below 60 mL/min/1.73m² potentially requiring 25-30% dose reductions to prevent accumulation. Liver function affects peptide metabolism through hepatic enzyme systems, though specific cytochrome P450 interactions with Thymagen remain poorly characterized in current literature. Concurrent immunosuppressive medications, including corticosteroids and methotrexate, may necessitate dose adjustments due to opposing effects on T-cell function.
Individual response variability demonstrates significant inter-subject differences, with some research participants showing optimal responses at 20-30 mcg while others require 60-80 mcg for comparable biomarker changes. Genetic polymorphisms affecting immune system function, particularly HLA typing and cytokine gene variants, contribute to this variability and may guide future personalized dosing approaches. Baseline immune status, including T-cell counts and immunoglobulin levels, influences optimal dosing strategies and response prediction.
What Happens If You Miss a Dose
Research protocols typically recommend administering missed Thymagen (Thymogen) doses within 12-24 hours of the scheduled time to maintain consistent plasma levels and immune system stimulation. If more than 24 hours have elapsed, protocols generally advise skipping the missed dose and resuming the regular schedule rather than doubling subsequent doses, which may cause immune system overstimulation or adverse reactions.
The peptide's short half-life of 2-4 hours means that missing doses by 48-72 hours effectively resets immune system stimulation, potentially requiring dose re-titration in sensitive research protocols. Studies examining dosing consistency demonstrate that irregular administration patterns reduce overall efficacy by 15-25% compared to consistent three-times-weekly schedules, emphasizing the importance of adherence to established protocols.
Dosing Compared to Similar Peptides
Thymagen (Thymogen) dosing differs significantly from other thymic peptides and immune modulators in both dose ranges and administration frequency. Compared to Thymosin Alpha-1, which typically requires 1.6-3.2 mg twice weekly, Thymagen utilizes much lower microgram doses due to its higher receptor binding affinity and different mechanism of action. The molecular weight difference (Thymagen: 278.3 Da vs. Thymosin Alpha-1: 3,108 Da) contributes to these dosing disparities and pharmacokinetic profiles.
| Peptide | Typical Dose | Frequency | Route | Half-Life | Key Difference |
|---|---|---|---|---|---|
| Thymagen (Thymogen) | 20-50 mcg | 3x weekly | Subcutaneous | 2-4 hours | Dipeptide, high potency |
| Thymosin Alpha-1 | 1.6-3.2 mg | 2x weekly | Subcutaneous | 2-3 hours | 28 amino acids, broader effects |
| Thymosin Beta-4 | 2-10 mg | 2x weekly | Subcutaneous | 1-2 hours | 43 amino acids, tissue repair |
| Epithalon | 5-10 mg | Daily | Subcutaneous | 30 minutes | Tetrapeptide, telomerase activation |
Common Dosing Mistakes
Starting with excessive doses represents the most frequent error in Thymagen (Thymogen) administration, with researchers sometimes beginning at 50-75 mcg without proper titration, leading to immune system overstimulation and skewed research results. Proper protocols require starting at 10-20 mcg to establish baseline responses and individual sensitivity patterns before escalating to higher doses over 2-4 week periods.
Inconsistent injection timing creates significant variability in plasma concentrations and immune system responses, with studies showing 20-30% reduced efficacy when administration times vary by more than 4-6 hours from scheduled doses. Research protocols emphasize maintaining consistent dosing intervals, typically every 48-72 hours for three-times-weekly schedules, to optimize peptide effects and data reliability.
Storage temperature violations compromise peptide stability and potency, with room temperature storage reducing Thymagen activity by 15-25% within 24 hours and causing complete degradation within 5-7 days. Proper storage at 2-8°C (36-46°F) maintains peptide integrity for 14-28 days post-reconstitution, while freezing temperatures (-20°C) can preserve lyophilized powder for 12-24 months.
Ignoring individual response markers leads to suboptimal dosing and poor research outcomes, as T-cell responses, cytokine levels, and immune function parameters vary significantly between subjects. Regular monitoring every 2-4 weeks allows dose optimization based on CD4+/CD8+ ratios, proliferation assays, and biomarker panels rather than following rigid protocols without adjustment.
Concurrent medication interactions, particularly with immunosuppressive drugs, corticosteroids, and certain antibiotics, can significantly alter Thymagen effectiveness and require dose modifications. Research protocols must account for these interactions and adjust dosing accordingly, often requiring 25-50% dose increases when used alongside immunosuppressive agents.
What the Evidence Does Not Show
Current research lacks comprehensive dose-response studies comparing different Thymagen (Thymogen) dosing regimens head-to-head in controlled clinical trials. While individual studies have examined specific dose ranges, no systematic analysis has determined optimal dosing for different research applications or subject populations, leaving significant gaps in evidence-based dosing recommendations.
Long-term safety data beyond 12-16 weeks of administration remains limited, with no studies examining chronic dosing effects on immune system function or potential adverse reactions from extended use. The maximum safe duration of Thymagen administration and appropriate washout periods between treatment cycles have not been established through rigorous clinical investigation.
Pediatric and geriatric dosing protocols lack specific evidence, as most research has focused on healthy adult subjects aged 18-65 years. Age-related pharmacokinetic changes, immune system differences, and safety considerations in vulnerable populations remain poorly characterized, limiting dosing guidance for these groups.
Combination dosing with other peptides or immunomodulatory agents has not been systematically studied, despite potential synergistic or antagonistic interactions that could significantly affect optimal dosing strategies. The lack of drug interaction studies limits clinical application and research protocol development involving multiple therapeutic agents.
Gender-specific dosing differences have not been adequately investigated, despite potential hormonal influences on immune system function and peptide metabolism that could affect optimal dose selection between male and female subjects.
FAQ
What is the standard dose of Thymagen (Thymogen)?
Research protocols typically employ 20-50 mcg administered subcutaneously three times weekly as standard dosing for most immune system studies. Starting doses of 10-20 mcg are recommended for initial assessment, with gradual titration to 30-75 mcg based on individual response markers and research objectives.[1]
How often do you take Thymagen (Thymogen)?
Most research protocols utilize three-times-weekly administration (Monday, Wednesday, Friday) to maintain consistent immune system stimulation while allowing adequate recovery time between doses. The peptide's 2-4 hour half-life necessitates frequent dosing to sustain therapeutic effects and optimal research outcomes.
Can you adjust the dose yourself?
Dose adjustments should only occur under qualified supervision with appropriate monitoring of immune system parameters including T-cell counts, cytokine levels, and biomarker panels. Self-adjustment without proper laboratory assessment can lead to immune system imbalances and compromise research validity or safety.
What time of day should you take it?
Morning administration on an empty stomach optimizes absorption and bioavailability, with most protocols recommending injection 30-60 minutes before breakfast. This timing prevents food interference with peptide absorption and allows for consistent plasma concentration profiles throughout research studies.
What if you miss a dose?
Administer missed doses within 12-24 hours of the scheduled time to maintain research protocol integrity. If more than 24 hours have elapsed, skip the missed dose and resume the regular schedule rather than doubling subsequent doses, which may cause immune system overstimulation.
Do men and women use the same dose?
Current research protocols typically use similar dosing ranges for both genders, though individual response variability may necessitate adjustments based on body weight, immune status, and biomarker responses. Gender-specific dosing guidelines have not been established through systematic clinical investigation.
How long should you take Thymagen (Thymogen)?
Research study durations typically range from 8-16 weeks, with most protocols employing 12-week treatment periods to assess immune system changes and biomarker responses. Long-term administration safety beyond 16 weeks has not been established through comprehensive clinical studies.
Is a higher dose more effective?
Dose-response relationships demonstrate optimal effects at 40-60 mcg doses for most research applications, with diminishing returns and potential adverse effects observed above 75-100 mcg per injection. Higher doses do not necessarily improve outcomes and may disrupt immune system balance.
How do you store reconstituted Thymagen (Thymogen)?
Store reconstituted peptide at 2-8°C (36-46°F) in the original vial, protected from light and contamination. The solution maintains stability for 14-28 days under proper storage conditions, with significant potency loss occurring at room temperature within 4-6 hours.
What needle size is recommended for injection?
Use 29-31 gauge insulin syringes with 0.5-1.0 mL capacity for precise subcutaneous administration. The small needle gauge minimizes injection site discomfort while ensuring accurate dose delivery and proper subcutaneous tissue penetration for optimal absorption.
This content is for informational purposes only and does not constitute medical advice. Thymagen (Thymogen) is available for research purposes only and is not FDA-approved for therapeutic use. Consult a licensed healthcare provider and qualified research supervisor before starting any treatment or research protocol.
References
- Ukrainskii biokhimicheskii zhurnal. "Effect of thymagen, thymalin and vilosen on the cAMP and cGMP levels and phosphodiesterase activity in spleen lymphocytes during sensitization and anaphylactic shock." 1991;63(4):54-58. PMID: 1659006