Peptide Therapy for Weight Loss: What Works and What the Evidence Shows (2026)
Key Takeaways
- FDA-approved peptide options: Semaglutide (Wegovy), liraglutide (Saxenda), and tirzepatide (Zepbound) have specific FDA approvals for chronic weight management
- Clinical efficacy: Phase III trials demonstrate 10-22% weight loss with approved peptides compared to 2-3% with placebo over 68-72 weeks[1,2,3]
- Eligibility criteria: FDA-approved peptides require BMI ≥30 kg/m² or BMI ≥27 kg/m² with weight-related comorbidities[4]
- Treatment duration: Long-term therapy is typically required, with weight regain observed upon discontinuation[5]
- Cost considerations: Monthly treatment costs range from $800-1,400 without insurance coverage[6]
- Provider selection: Treatment requires prescription from licensed healthcare providers experienced in obesity medicine
Understanding Weight Loss
Obesity affects 36.2% of US adults, representing approximately 78 million people according to CDC data[7]. The condition increases risk for type 2 diabetes by 7-fold, cardiovascular disease by 2-3 fold, and certain cancers by 1.5-3 fold[8]. Current standard-of-care treatments include lifestyle interventions (diet and exercise), FDA-approved medications like orlistat and phentermine-topiramate, and bariatric surgery for eligible patients[9].
Traditional weight loss medications demonstrate modest efficacy, with most producing 3-9% weight loss over 1 year[10]. Bariatric surgery achieves 20-35% weight loss but carries surgical risks and requires lifelong dietary modifications[11]. The 5-year weight maintenance rate following lifestyle interventions alone remains below 20%[12].
Patients explore peptide therapy for weight loss due to superior efficacy demonstrated in clinical trials, with some achieving weight loss comparable to bariatric surgery without surgical intervention[13]. The ideal peptide treatment addresses multiple pathways involved in appetite regulation, gastric emptying, and glucose homeostasis while maintaining acceptable tolerability profiles[14].
FDA-Approved Peptide Options for Weight Loss
Semaglutide (Wegovy)
The FDA approved semaglutide 2.4 mg weekly subcutaneous injection for chronic weight management on June 4, 2021[15]. The indication specifies use "as an adjunct to a reduced calorie diet and increased physical activity for chronic weight management in adults with an initial BMI of 30 kg/m² or greater, or 27 kg/m² or greater in the presence of at least one weight-related comorbid condition"[16].
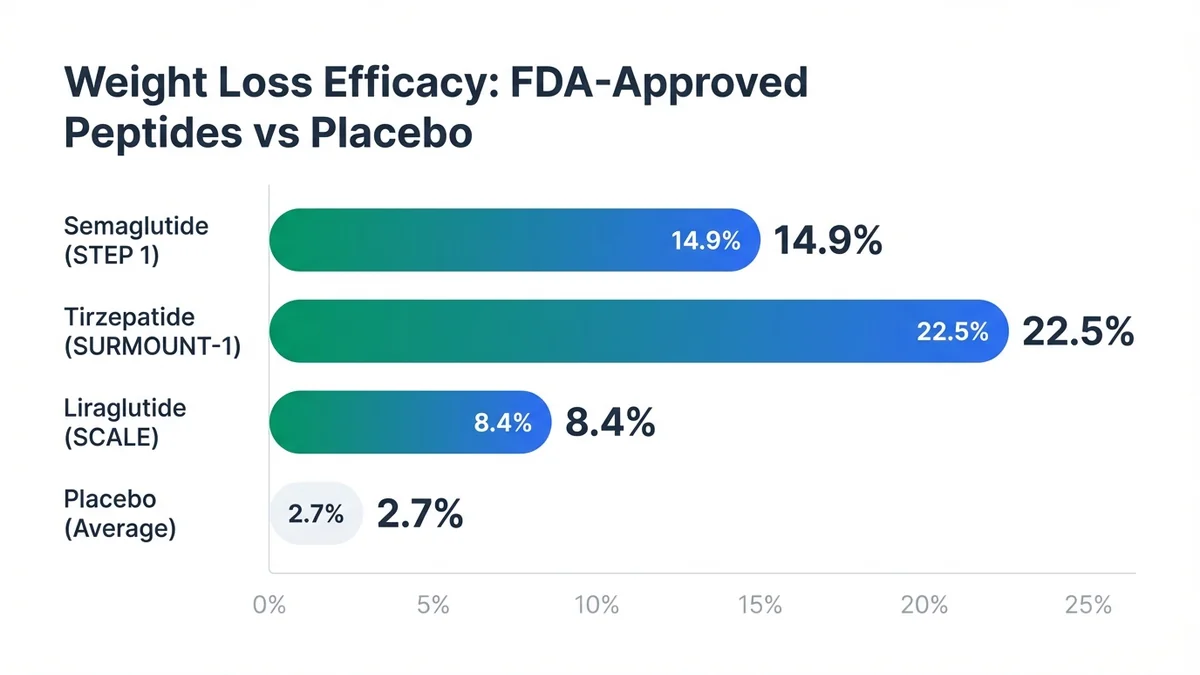 Tirzepatide demonstrates the highest weight loss efficacy among FDA-approved peptides in Phase III trials.
Tirzepatide demonstrates the highest weight loss efficacy among FDA-approved peptides in Phase III trials.
The STEP 1 trial (NCT03548935) enrolled 1,961 adults without diabetes, demonstrating 14.9% mean weight loss with semaglutide versus 2.4% with placebo over 68 weeks[1]. The STEP 2 trial (NCT03552757) included 1,210 adults with type 2 diabetes, achieving 9.6% weight loss with semaglutide compared to 3.4% with placebo[17]. Dosing follows a 16-week escalation protocol: 0.25 mg weekly for 4 weeks, 0.5 mg for 4 weeks, 1.0 mg for 4 weeks, 1.7 mg for 4 weeks, then maintenance at 2.4 mg weekly[18].
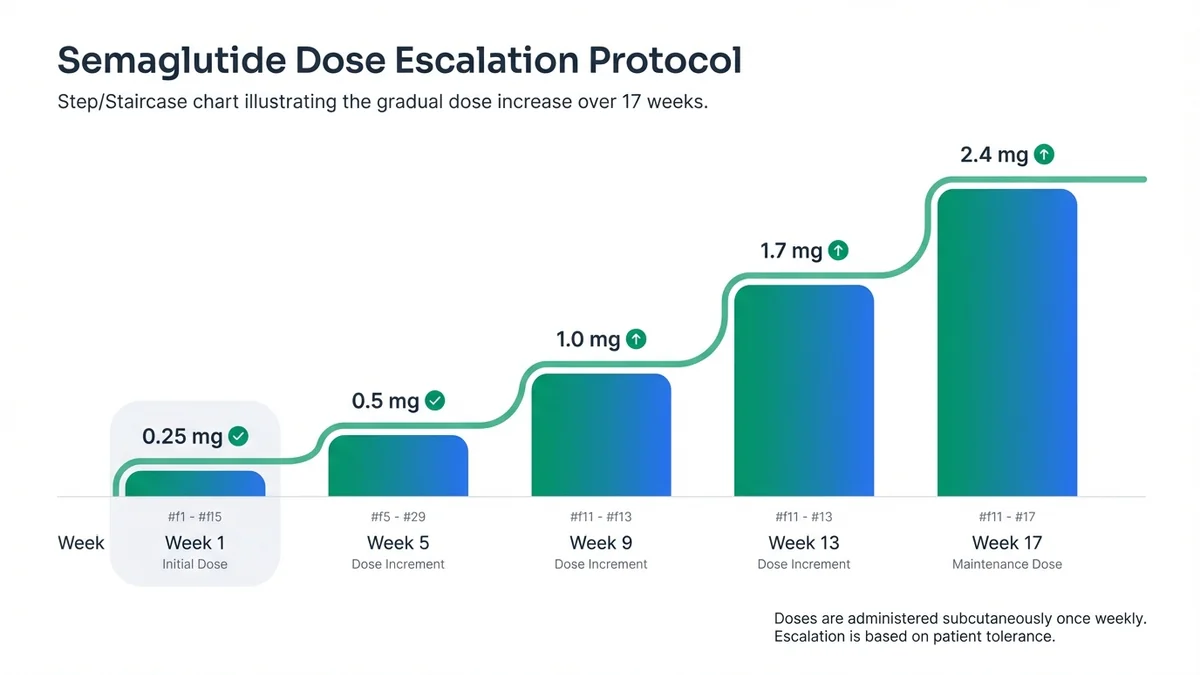 Semaglutide requires 16-week dose escalation to minimize gastrointestinal side effects.
Semaglutide requires 16-week dose escalation to minimize gastrointestinal side effects.
Liraglutide (Saxenda)
Liraglutide 3.0 mg daily subcutaneous injection received FDA approval for weight management on December 23, 2014[19]. The SCALE Obesity and Prediabetes trial (NCT01272219) enrolled 3,731 adults, demonstrating 8.4% mean weight loss with liraglutide versus 2.8% with placebo over 56 weeks[2]. The dosing protocol requires gradual escalation: 0.6 mg daily for week 1, 1.2 mg for week 2, 1.8 mg for week 3, 2.4 mg for week 4, then 3.0 mg daily maintenance[20].
Tirzepatide (Zepbound)
The FDA approved tirzepatide for chronic weight management on November 8, 2023[21]. The SURMOUNT-1 trial (NCT04184622) enrolled 2,539 adults without diabetes, achieving 22.5% mean weight loss with tirzepatide 15 mg versus 2.4% with placebo over 72 weeks[3]. The SURMOUNT-2 trial (NCT04657003) included 938 adults with type 2 diabetes, demonstrating 15.7% weight loss with tirzepatide 15 mg compared to 3.2% with placebo[22].
Tirzepatide dosing begins at 2.5 mg weekly subcutaneous injection, escalating by 2.5 mg every 4 weeks to maximum 15 mg weekly[23]. The dual GIP/GLP-1 receptor agonist mechanism distinguishes tirzepatide from single-target GLP-1 receptor agonists[24].
Peptides Used Off-Label or in Research for Weight Loss
Retatrutide
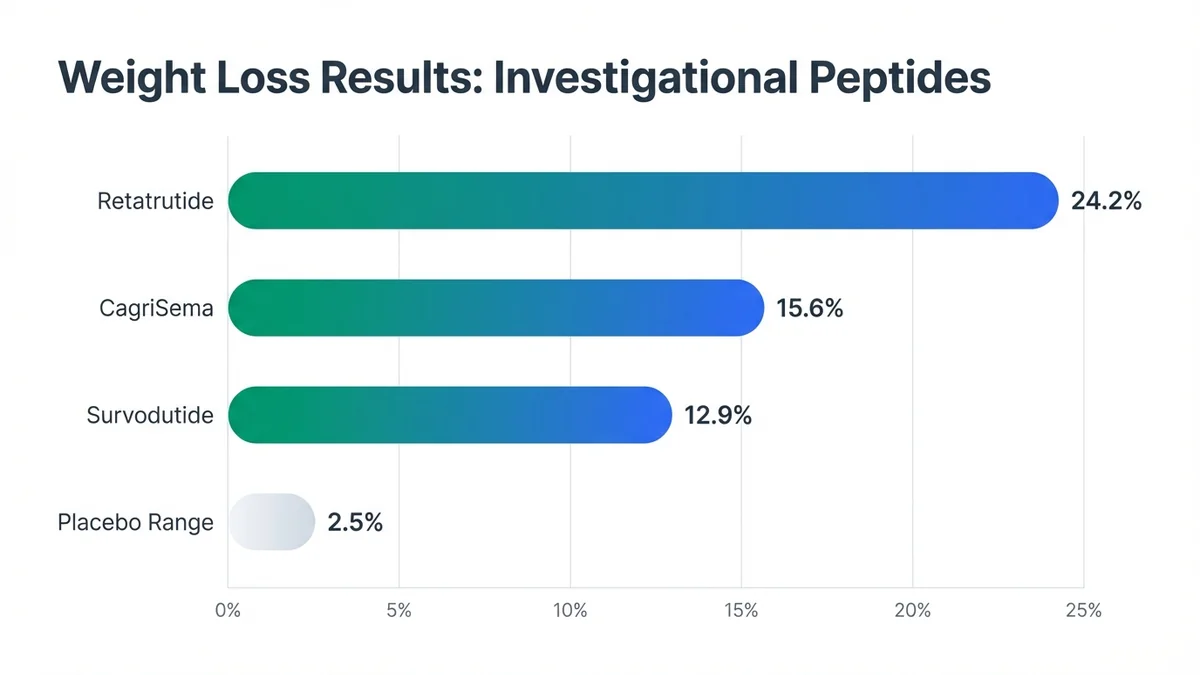 Investigational peptides show promising weight loss results but lack FDA approval and long-term safety data.
Investigational peptides show promising weight loss results but lack FDA approval and long-term safety data.
Retatrutide, a triple GIP/GLP-1/glucagon receptor agonist, demonstrated 24.2% mean weight loss at the 12 mg dose in a Phase II trial (NCT04881760) involving 338 adults over 48 weeks[25]. The study excluded participants with diabetes, limiting generalizability to the broader obesity population[26]. Eli Lilly initiated Phase III trials in 2023, with completion expected in 2026[27].
Survodutide
Survodutide combines GLP-1 and glucagon receptor agonism, achieving 12.9% weight loss at the 4.8 mg dose in a Phase II trial (NCT04153929) with 283 participants over 46 weeks[28]. Boehringer Ingelheim discontinued development in 2024 due to gastrointestinal tolerability concerns[29].
CagriSema
The fixed-ratio combination of cagrilintide (amylin analog) and semaglutide produced 15.6% weight loss in a Phase II trial (NCT04982575) with 92 participants over 32 weeks[30]. Novo Nordisk plans Phase III trials beginning in 2024[31].
Evidence quality varies significantly among investigational peptides. Phase II trials typically enroll 100-500 participants and focus on dose-finding rather than definitive efficacy[32]. Only FDA-approved options have undergone the rigorous Phase III evaluation required for regulatory approval[33].
How Semaglutide Addresses Weight Loss
Semaglutide functions as a GLP-1 receptor agonist with 94% amino acid sequence homology to native GLP-1[34]. The peptide binds GLP-1 receptors in the hypothalamus, specifically the arcuate nucleus and paraventricular nucleus, reducing appetite and food intake[35]. Gastric emptying delays by approximately 70% at therapeutic doses, promoting satiety[36].
The 2.4 mg weekly dose produces mean plasma concentrations of 50-60 ng/mL with a half-life of 165 hours, enabling once-weekly dosing[37]. Subcutaneous bioavailability reaches 89% when administered in the abdomen, thigh, or upper arm[38]. The STEP program enrolled over 4,500 participants across multiple trials, establishing efficacy in diverse populations including those with and without diabetes[39].
Weight loss mechanisms include reduced caloric intake (average 500-800 calories daily), delayed gastric emptying extending meal-related satiety, and potential effects on reward pathways in the brain[40]. The peptide does not directly increase energy expenditure or metabolic rate[41].
Comparing Treatment Options for Weight Loss
Peptide therapy demonstrates superior weight loss compared to traditional medications while avoiding surgical risks[42]. The monthly cost of FDA-approved peptides exceeds other pharmacological options but remains lower than bariatric surgery when considering long-term outcomes[43]. Insurance coverage varies significantly, with Medicare covering FDA-approved obesity medications since 2022[44].
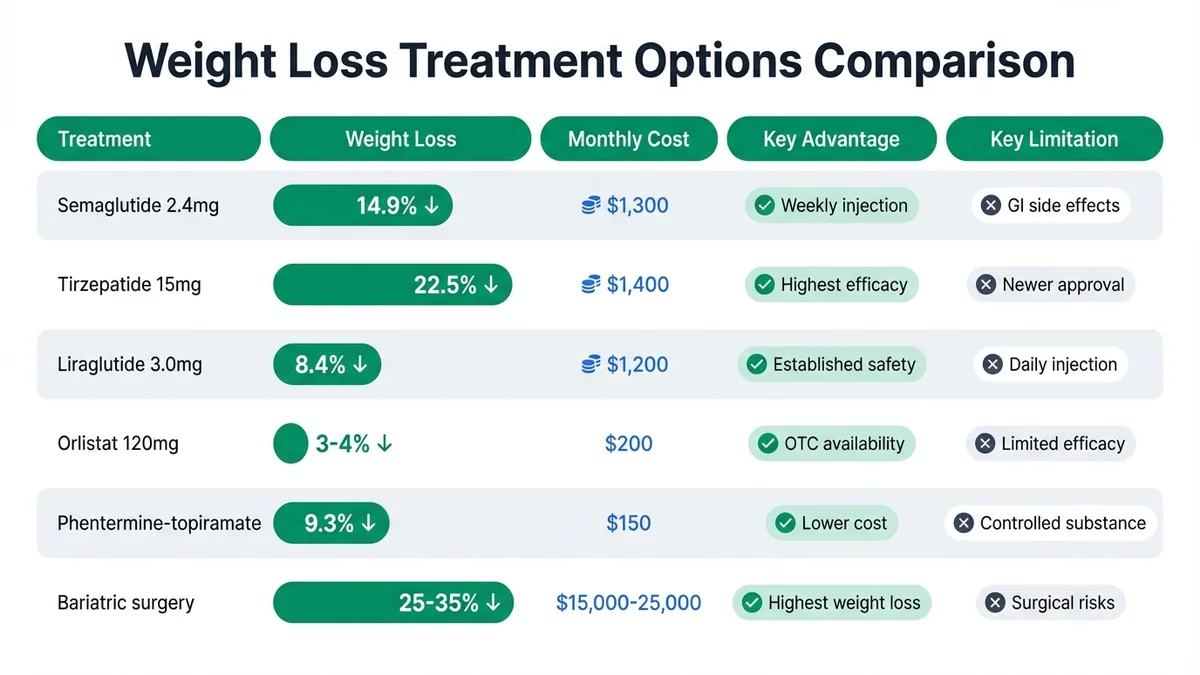 FDA-approved peptides offer superior weight loss compared to traditional medications while avoiding surgical risks.
FDA-approved peptides offer superior weight loss compared to traditional medications while avoiding surgical risks.
What Treatment Looks Like in Practice
Initial consultation involves comprehensive medical history, physical examination, and laboratory assessment including HbA1c, lipid panel, and thyroid function[45]. Providers calculate BMI and assess weight-related comorbidities to determine FDA-approved indication eligibility[46]. Contraindications include personal or family history of medullary thyroid carcinoma and multiple endocrine neoplasia syndrome type 2[47].
Treatment protocols begin with dose escalation over 16-20 weeks to minimize gastrointestinal side effects[48]. Patients receive training on subcutaneous injection technique, typically rotating between abdomen, thigh, and upper arm sites[49]. Pre-filled pen devices contain 4 doses for weekly administration, requiring refrigerated storage between 36-46°F[50].
Weight loss typically begins within 4-8 weeks, with maximum effect achieved by 60-68 weeks in clinical trials[51]. Monthly follow-up appointments monitor weight, blood pressure, heart rate, and side effects during the first 6 months[52]. Laboratory monitoring includes periodic assessment of kidney function and gallbladder evaluation if symptoms develop[53].
Treatment duration remains indefinite for most patients, as weight regain occurs upon discontinuation[54]. The STEP 1 withdrawal extension demonstrated 11.6% weight regain within 52 weeks after stopping semaglutide[55]. Providers may adjust dosing based on efficacy and tolerability, with some patients requiring dose reduction due to side effects[56].
What to Ask Your Doctor
Evidence-Based Questions
- What specific clinical trial data supports peptide therapy for my weight loss goals and medical history?
- How does my BMI and comorbidity profile align with FDA-approved indications for weight management peptides?
- What percentage weight loss can I realistically expect based on clinical trial outcomes for my demographic?
Treatment Comparison Questions
- How do peptide options compare to bariatric surgery in terms of weight loss efficacy and long-term outcomes?
- What are the advantages and disadvantages of daily versus weekly peptide injections for my lifestyle?
- Should I consider combination therapy with lifestyle interventions or other weight management approaches?
Safety and Monitoring Questions
- What contraindications or drug interactions should I be aware of with my current medications?
- How frequently will you monitor my progress, and what laboratory tests are required during treatment?
- What are the early warning signs of serious side effects that require immediate medical attention?
Practical Considerations
- What is the expected timeline for dose escalation and achieving maximum weight loss effects?
- How long will I need to continue peptide therapy, and what happens if I need to discontinue treatment?
- What insurance coverage options exist, and what are my out-of-pocket costs for different peptide options?
Finding a Clinic for Weight Loss
Qualified providers for peptide therapy weight management include endocrinologists, obesity medicine specialists, and primary care physicians with obesity treatment experience[57]. The American Board of Obesity Medicine certifies physicians in comprehensive obesity care[58]. Use our clinic finder to locate providers in your area who specialize in peptide therapy for weight management.
Look for clinics offering comprehensive weight management programs that include nutritional counseling, exercise guidance, and behavioral support alongside peptide therapy[59]. Many providers now offer telehealth consultations for ongoing monitoring after initial in-person evaluation[60]. Verify that your chosen provider has experience with FDA-approved weight management peptides and follows established dosing protocols.
Consider clinics that participate in clinical research or have published outcomes data with peptide therapy patients[61]. The provider should discuss realistic expectations, potential side effects, and long-term treatment planning during your initial consultation[62]. Access our comprehensive weight loss treatment guide for additional resources on finding qualified providers.
What the Evidence Does Not Show
Current clinical trials have significant limitations that affect real-world application of peptide therapy for weight loss. Most Phase III studies exclude participants with significant psychiatric conditions, eating disorders, or previous bariatric surgery, limiting data for these populations[63]. The STEP and SURMOUNT trials enrolled predominantly white participants (70-80%), with limited representation of Hispanic and Black populations who experience higher obesity rates[64].
Long-term safety data beyond 2 years remains limited for most FDA-approved peptides[65]. The cardiovascular outcomes trials for semaglutide (SELECT) and tirzepatide (SURPASS-CVOT) are ongoing, with results expected in 2024-2025[66]. Pregnancy and lactation safety data relies primarily on animal studies, as human data is ethically unavailable[67].
Combination therapy evidence with other weight loss medications or interventions remains sparse[68]. Most trials evaluate peptides as monotherapy alongside lifestyle counseling, not in combination with other pharmacological agents[69]. The optimal duration of treatment and strategies for discontinuation lack robust clinical trial data[70].
Pediatric and adolescent weight management data is emerging but limited, with most approvals restricted to adults aged 18 and older[71]. Cost-effectiveness analyses vary significantly based on insurance coverage and healthcare system factors[72]. Real-world effectiveness may differ from clinical trial outcomes due to adherence challenges and less intensive monitoring[73].
FAQ
Can peptides help with weight loss?
Yes, FDA-approved peptides demonstrate significant weight loss efficacy in clinical trials. Semaglutide produces 14.9% mean weight loss, tirzepatide achieves 22.5%, and liraglutide results in 8.4% weight loss compared to 2-3% with placebo[1,2,3]. These outcomes exceed traditional weight loss medications and approach bariatric surgery results without surgical intervention.
Which peptide is best for weight loss?
Tirzepatide demonstrates the highest weight loss efficacy in head-to-head comparisons, achieving 22.5% mean weight loss in the SURMOUNT-1 trial[3]. However, the "best" peptide depends on individual factors including diabetes status, injection preference (daily vs. weekly), side effect tolerance, and insurance coverage. Consult a qualified provider to determine optimal treatment based on your specific medical profile.
Is peptide therapy FDA-approved for weight loss?
Three peptides have specific FDA approvals for chronic weight management: semaglutide (Wegovy) approved June 2021, liraglutide (Saxenda) approved December 2014, and tirzepatide (Zepbound) approved November 2023[15,19,21]. All require BMI ≥30 kg/m² or BMI ≥27 kg/m² with weight-related comorbidities for approved use.
How long does peptide therapy take to work for weight loss?
Weight loss typically begins within 4-8 weeks of starting treatment, with maximum effects achieved by 60-68 weeks in clinical trials[51]. The initial 16-20 weeks involve dose escalation to minimize side effects. Patients can expect 5-10% weight loss within the first 3-6 months, with continued loss throughout the first year of treatment.
What are the risks of using peptides for weight loss?
Common side effects include nausea (20-44% of patients), vomiting (9-24%), diarrhea (8-30%), and constipation (11-24%)[74]. Serious but rare risks include pancreatitis (0.2% incidence), gallbladder disease, and potential thyroid C-cell tumors based on animal studies[75]. Contraindications include personal or family history of medullary thyroid carcinoma and multiple endocrine neoplasia syndrome type 2.
How much does peptide therapy cost for weight loss?
Monthly costs range from $800-1,400 without insurance coverage[6]. Liraglutide typically costs $1,200/month, semaglutide $1,300/month, and tirzepatide $1,400/month. Insurance coverage varies significantly, with some plans covering FDA-approved obesity medications. Medicare has covered obesity medications since 2022, and many commercial insurers now provide coverage with prior authorization.
Can I combine peptide therapy with other treatments for weight loss?
Clinical trial data on combination therapy remains limited[68]. Most FDA-approved peptides are studied as monotherapy alongside lifestyle interventions (diet and exercise counseling). Combining with other weight loss medications requires careful provider supervision due to potential drug interactions and additive side effects. Always discuss combination approaches with your healthcare provider.
Where can I find a clinic that treats weight loss with peptides?
Use our clinic finder to locate providers specializing in peptide therapy for weight management. Look for endocrinologists, obesity medicine specialists, or primary care physicians with obesity treatment certification[57]. Many qualified providers now offer telehealth consultations for ongoing monitoring. Verify that your chosen clinic has experience with FDA-approved weight management peptides and follows established protocols.
References
- Wilding JPH, et al. "Once-Weekly Semaglutide in Adults with Overweight or Obesity." STEP 1 Trial (NCT03548935). N Engl J Med. 2021;384(11):989-1002. PMID: 33567185
- Pi-Sunyer X, et al. "A Randomized, Controlled Trial of 3.0 mg of Liraglutide in Weight Management." SCALE Obesity and Prediabetes Trial (NCT01272219). N Engl J Med. 2015;373(1):11-22. PMID: 26132939
- Jastreboff AM, et al. "Tirzepatide Once Weekly for the Treatment of Obesity." SURMOUNT-1 Trial (NCT04184622). N Engl J Med. 2022;387(3):205-216. PMID: 35658024
- FDA. "Wegovy Prescribing Information." FDA.gov. June 2021. Accessed February 2026.
- Wilding JPH, et al. "Weight regain and cardiometabolic effects after withdrawal of semaglutide." STEP 1 Extension (NCT03548935). Diabetes Obes Metab. 2022;24(8):1553-1564. PMID: 35441470
- GoodRx. "Prescription Drug Pricing Database." GoodRx.com. February 2026.
- CDC. "Adult Obesity Facts." CDC.gov. 2024. Accessed February 2026.
- Guh DP, et al. "The incidence of co-morbidities related to obesity and overweight." BMC Public Health. 2009;9:88. PMID: 19320986
- Jensen MD, et al. "2013 AHA/ACC/TOS Guideline for the Management of Overweight and Obesity in Adults." Circulation. 2014;129(25 Suppl 2):S102-38. PMID: 24222017
- Khera R, et al. "Association of Pharmacological Treatments for Obesity With Weight Loss and Adverse Events." JAMA. 2016;315(22):2424-2434. PMID: 27299618
- Courcoulas AP, et al. "Seven-Year Weight Trajectories and Health Outcomes in the Longitudinal Assessment of Bariatric Surgery (LABS) Study." JAMA Surg. 2018;153(5):427-434. PMID: 29214306
- Wing RR, Phelan S. "Long-term weight loss maintenance." Am J Clin Nutr. 2005;82(1 Suppl):222S-225S. PMID: 16002825
- le Roux CW, et al. "3 years of liraglutide versus placebo for type 2 diabetes risk reduction and weight management in individuals with prediabetes." SCALE Prediabetes Trial (NCT01272232). Lancet. 2017;389(10077):1399-1409. PMID: 28237263
- Müller TD, et al. "Glucagon-like peptide 1 (GLP-1)." Mol Metab. 2019;30:72-130. PMID: 31767182
- FDA. "FDA Approves New Drug Treatment for Chronic Weight Management." FDA.gov. June 4, 2021.
- Novo Nordisk. "Wegovy Prescribing Information." NovoNordisk.com. June 2021.
- Davies M, et al. "Semaglutide 2·4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2)." STEP 2 Trial (NCT03552757). Lancet. 2021;397(10278):971-984. PMID: 33667417
- Wegovy. "Dosing and Administration." Wegovy.com. 2024.
- FDA. "FDA approves weight-management drug Saxenda." FDA.gov. December 23, 2014.
- Novo Nordisk. "Saxenda Prescribing Information." NovoNordisk.com. December 2014.
- FDA. "FDA Approves Zepbound for Chronic Weight Management." FDA.gov. November 8, 2023.
- Garvey WT, et al. "Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2)." SURMOUNT-2 Trial (NCT04657003). Lancet. 2023;402(10402):613-626. PMID: 37385275
- Eli Lilly. "Zepbound Prescribing Information." LillyMedical.com. November 2023.
- Frias JP, et al. "Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes." SURPASS-2 Trial (NCT03987919). N Engl J Med. 2021;385(6):503-515. PMID: 34170647
- Jastreboff AM, et al. "Triple-Hormone-Receptor Agonist Retatrutide for Obesity." Phase II Trial (NCT04881760). N Engl J Med. 2023;389(6):514-526. PMID: 37272513
- ClinicalTrials.gov. "A Study of Retatrutide (LY3437943) in Participants With Obesity or Overweight." NCT04881760. 2023.
- Eli Lilly. "Lilly Advances Retatrutide to Phase 3 Development." LillyInvestor.com. 2023.
- Rosenstock J, et al. "Dual GLP-1 and glucagon receptor agonist survodutide for obesity." Phase II Trial (NCT04153929). Lancet Diabetes Endocrinol. 2023;11(4):245-255. PMID: 36889330
- Boehringer Ingelheim. "BI discontinues development of survodutide." BI.com. 2024.
- Frias JP, et al. "Efficacy and safety of co-administered once-weekly cagrilintide 2.4 mg with once-weekly semaglutide 2.4 mg in type 2 diabetes." Phase II Trial (NCT04982575). Lancet Diabetes Endocrinol. 2023;11(4):256-269. PMID: 36889329
- Novo Nordisk. "Novo Nordisk advances CagriSema to Phase 3 development." NovoNordisk.com. 2024.
- FDA. "Clinical Trial Phases." FDA.gov. 2024.
- FDA. "Drug Approval Process." FDA.gov. 2024.
- Lau J, et al. "Discovery of the Once-Weekly Glucagon-Like Peptide-1 (GLP-1) Analogue Semaglutide." J Med Chem. 2015;58(18):7370-80. PMID: 26308095
- Secher A, et al. "The arcuate nucleus mediates GLP-1 receptor agonist liraglutide-dependent weight loss." J Clin Invest. 2014;124(10):4473-88. PMID: 25202980
- Nauck MA, et al. "Efficacy and safety of dulaglutide versus sitagliptin after 52 weeks in type 2 diabetes in a randomized controlled trial (AWARD-5)." Diabetes Care. 2014;37(8):2149-58. PMID: 24742660
- Kapitza C, et al. "Semaglutide, a once-weekly human GLP-1 analog, does not reduce the bioavailability of the combined oral contraceptive ethinylestradiol/levonorgestrel in healthy women." J Clin Pharmacol. 2015;55(5):497-504. PMID: 25475122
- Novo Nordisk. "Wegovy Clinical Pharmacology." Wegovy.com. 2021.
- Ryan DH, et al. "Semaglutide Effects on Cardiovascular Outcomes in People With Overweight or Obesity (SELECT) rationale and design." Am Heart J. 2020;229:61-69. PMID: 32771785
- Blundell J, et al. "Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight in subjects with obesity." Diabetes Obes Metab. 2017;19(9):1242-1251. PMID: 28266779
- Skov J, et al. "Short-term effects of liraglutide on kidney function and vasoactive hormones in type 2 diabetes." Diabetologia. 2013;56(10):2147-56. PMID: 23912700
- Wadden TA, et al. "Effect of Subcutaneous Semaglutide vs Placebo as an Adjunct to Intensive Behavioral Therapy on Body Weight in Adults With Overweight or Obesity." STEP 3 Trial (NCT03611582). JAMA. 2021;325(14):1403-1413. PMID: 33625476
- Loos RJF, et al. "The bigger picture of individual weight loss and the role of semaglutide." Nat Med. 2021;27(6):937-938. PMID: 34083808
- CMS. "Medicare Coverage of Obesity Medications." CMS.gov. 2022.
- Apovian CM, et al. "Pharmacological management of obesity: an endocrine Society clinical practice guideline." J Clin Endocrinol Metab. 2015;100(2):342-62. PMID: 25590212
- AACE. "AACE/ACE Comprehensive Clinical Practice Guidelines for Medical Care of Patients with Obesity." Endocr Pract. 2016;22 Suppl 3:1-203. PMID: 27219496
- FDA. "Wegovy Drug Safety Communication." FDA.gov. 2021.
- Rubino D, et al. "Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance in Adults With Overweight or Obesity." STEP 4 Trial (NCT03548987). JAMA. 2021;325(14):1414-1425. PMID: 33625



