Peptide Therapy Clinics: Your Top Questions Answered by Experts (2026)
Key Takeaways
- Peptide therapy clinics must employ licensed physicians and use FDA-approved compounding pharmacies to ensure patient safety and regulatory compliance[1]
- Legitimate clinics require comprehensive lab work (typically 15-20 biomarkers) before prescribing any peptide therapy[2]
- Monthly monitoring visits are standard practice, with telehealth options available in 47 states as of 2026[3]
- Treatment costs range from $200-800 per month depending on peptide type, with most insurance plans not covering off-label peptide use[4]
- Board-certified physicians in endocrinology, anti-aging medicine, or sports medicine typically have the most relevant training for peptide therapy[5]
- Red flags include clinics offering peptides without prescriptions, using non-FDA registered pharmacies, or making disease cure claims[6]
About Peptide Therapy Clinics
Peptide therapy clinics specialize in prescribing bioactive peptides for various health optimization and medical purposes. These facilities operate under strict FDA regulations, as peptides are classified as prescription medications when used for therapeutic purposes[7]. Licensed physicians at these clinics prescribe peptides through 503A compounding pharmacies, which must follow FDA guidelines for sterile compounding and quality control[8].
The peptide therapy field encompasses over 60 different therapeutic peptides, ranging from FDA-approved medications like semaglutide for diabetes and weight management to research peptides like BPC-157 for tissue repair[9]. Clinics typically focus on specific therapeutic areas such as weight management, anti-aging, athletic performance, or hormone optimization, requiring specialized knowledge of peptide pharmacokinetics and patient monitoring protocols[10].
Frequently Asked Questions
What qualifications should a peptide therapy clinic have?
A legitimate peptide therapy clinic must employ licensed physicians with DEA prescribing authority and maintain partnerships with FDA-registered 503A compounding pharmacies[11]. Board certification in relevant specialties such as endocrinology, anti-aging medicine (A4M), sports medicine, or family medicine provides the clinical foundation for peptide prescribing[12]. The clinic should display state medical licenses, DEA registration numbers, and pharmacy partnership documentation transparently on their website or upon request[13].
Additional credentials include membership in professional organizations like the American Academy of Anti-Aging Medicine (A4M) or the International Peptide Society, which require continuing education in peptide therapeutics[14]. Many reputable clinics also maintain accreditation through organizations like the Accreditation Association for Ambulatory Health Care (AAAHC), which conducts on-site inspections of clinical protocols and safety procedures[15].
How do I verify a clinic's FDA compliance?
Verify FDA compliance by confirming the clinic uses only FDA-registered 503A compounding pharmacies, which you can check through the FDA's online database of registered facilities[16]. Request documentation showing the pharmacy's FDA registration number, state licensing, and most recent inspection reports[17]. Legitimate clinics will provide this information readily, as they understand patient safety depends on pharmaceutical quality control[18].
The clinic should also follow FDA guidelines for off-label prescribing, which requires documented medical necessity and informed consent procedures[19]. Ask to review their informed consent forms, which should detail specific peptide risks, benefits, and alternative treatments according to FDA guidance on off-label drug use[20]. You can find qualified, compliant clinics through our clinic directory, which verifies these credentials before listing providers.
What lab work is required before starting peptide therapy?
Comprehensive lab work typically includes 15-20 biomarkers depending on the specific peptide therapy being considered[21]. For metabolic peptides like tirzepatide, required tests include hemoglobin A1C, fasting glucose, lipid panel, kidney function (creatinine, eGFR), liver enzymes (ALT, AST), and thyroid function (TSH, T3, T4)[22]. Growth hormone-related peptides require additional testing for IGF-1 levels, complete blood count, and cardiac markers[23].
Most clinics require lab work within 30-90 days of treatment initiation, with follow-up testing every 3-6 months during active treatment[24]. The specific panel varies by peptide class: cardiovascular peptides require lipid profiles and inflammatory markers (CRP, homocysteine), while cognitive enhancement peptides may include neurotransmitter metabolites and B-vitamin levels[25]. Expect to pay $200-500 for comprehensive initial lab work, though some clinics include this in their consultation fees[26].
How much does peptide therapy cost at clinics?
Monthly peptide therapy costs range from $200-800 depending on the specific peptide, dosage, and clinic pricing structure[27]. FDA-approved peptides like semaglutide typically cost $300-500 monthly through specialized clinics, compared to $1,000+ through traditional pharmacies without insurance coverage[28]. Research peptides and combination therapies generally fall in the $400-800 range due to specialized compounding requirements[29].
Additional costs include initial consultations ($150-300), required lab work ($200-500), and follow-up visits ($100-200 monthly)[30]. Many clinics offer package deals combining consultation, labs, and 3-month peptide supplies for $1,200-2,000[31]. Insurance rarely covers off-label peptide use, though HSA and FSA accounts can typically be used for legitimate medical treatments prescribed by licensed physicians[32]. Compare pricing and services using our cost comparison tool.
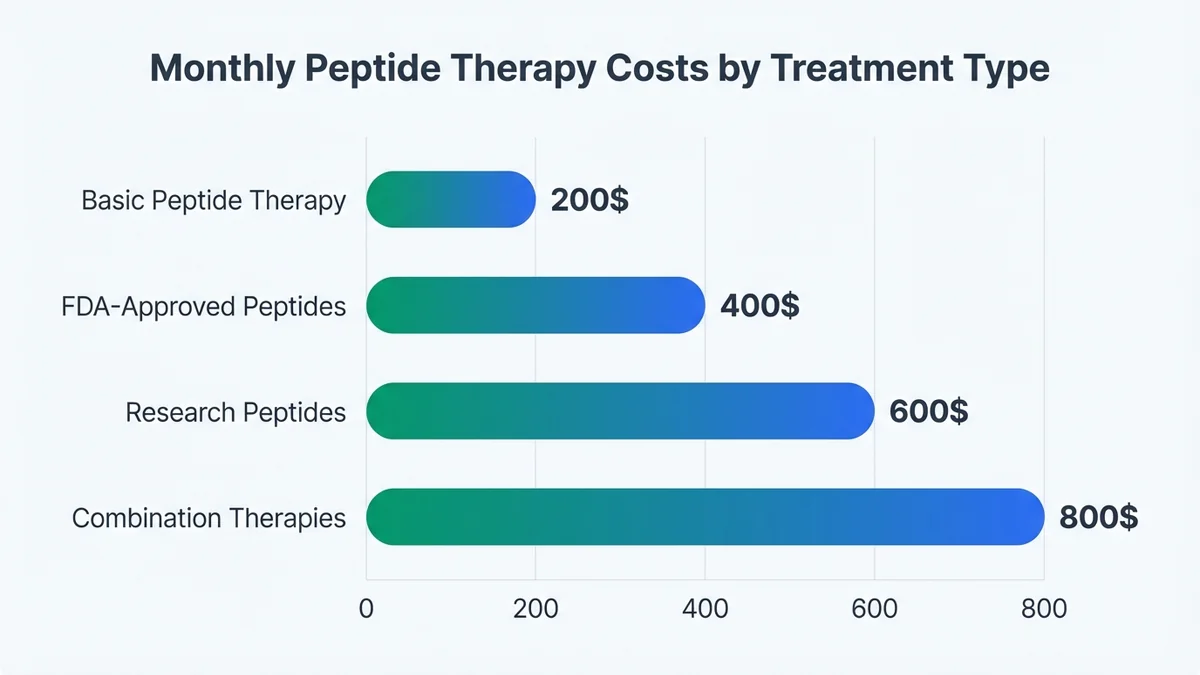 Monthly peptide therapy costs vary significantly by peptide type and clinic pricing structure.
Monthly peptide therapy costs vary significantly by peptide type and clinic pricing structure.
What's the difference between telehealth and in-person peptide clinics?
Telehealth peptide clinics operate in 47 states as of 2026, offering virtual consultations with licensed physicians who can prescribe peptides and coordinate lab work through local facilities[33]. These clinics typically charge 20-30% less than in-person facilities due to lower overhead costs, with consultation fees ranging from $100-200 versus $200-300 for office visits[34]. Telehealth providers must maintain licenses in each state where they treat patients and follow state-specific telemedicine regulations[35].
In-person clinics offer direct injection training, immediate side effect assessment, and hands-on monitoring that some patients prefer for complex protocols[36]. Physical clinics also provide additional services like IV therapy, body composition analysis, and comprehensive wellness programs that complement peptide treatment[37]. Both delivery methods require the same physician oversight and pharmacy compliance, though in-person clinics may offer more intensive monitoring for high-risk patients[38]. Explore both options through our telehealth directory and local clinic finder.
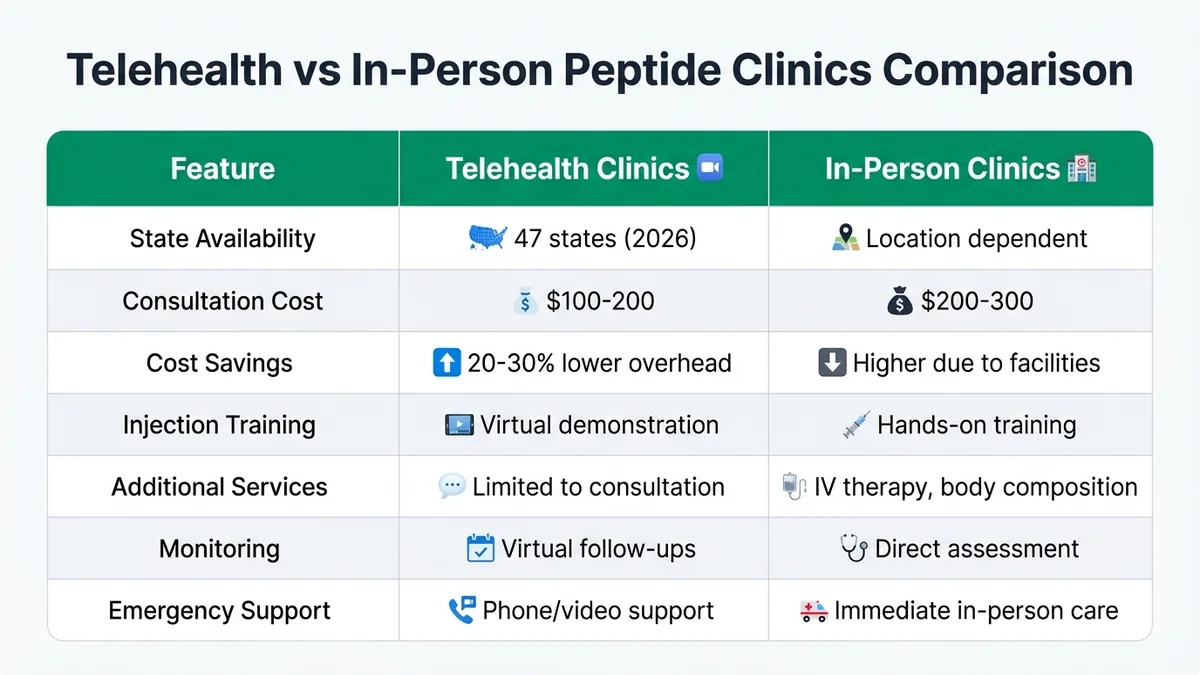 Telehealth clinics offer cost savings and convenience while in-person clinics provide hands-on training and comprehensive services.
Telehealth clinics offer cost savings and convenience while in-person clinics provide hands-on training and comprehensive services.
How do I know if a peptide clinic is legitimate?
Legitimate peptide clinics display current medical licenses, DEA registration numbers, and maintain partnerships with FDA-registered 503A compounding pharmacies[39]. Verify physician credentials through your state medical board database, which lists license status, board certifications, and any disciplinary actions[40]. The clinic should require comprehensive medical history, current medication review, and appropriate lab work before prescribing any peptide therapy[41].
Warning signs include clinics selling peptides without prescriptions, using research chemical suppliers instead of licensed pharmacies, or making disease cure claims prohibited by FDA regulations[42]. Legitimate providers will discuss both benefits and risks transparently, provide detailed informed consent documents, and maintain regular follow-up schedules[43]. Check clinic reviews and Better Business Bureau ratings, but focus primarily on verifiable credentials and regulatory compliance[44].
What questions should I ask during a peptide clinic consultation?
Ask about the physician's specific training in peptide therapy, including board certifications and continuing education in hormone optimization or anti-aging medicine[45]. Request details about their compounding pharmacy partnerships, including FDA registration numbers and quality control procedures[46]. Inquire about their patient monitoring protocols, including frequency of follow-up visits, required lab work intervals, and emergency contact procedures[47].
Essential questions include: "What specific lab work do you require before prescribing?" "How do you monitor for side effects?" "What is your protocol if I experience adverse reactions?" and "Can you provide references from your compounding pharmacy?"[48] Ask about total treatment costs, insurance coverage options, and their policy for discontinuing treatment[49]. Document their responses and compare answers across multiple clinics to identify the most thorough and transparent providers[50].
How long does it take to see results from peptide therapy?
Results timelines vary significantly by peptide type and individual patient factors, with most therapeutic effects becoming apparent within 4-12 weeks of consistent treatment[51]. Weight management peptides like semaglutide typically show initial appetite suppression within 1-2 weeks, with significant weight loss (5-10%) occurring over 12-16 weeks[52]. Growth hormone-releasing peptides may improve sleep quality within 2-4 weeks, with body composition changes becoming measurable after 8-12 weeks[53].
Cognitive enhancement peptides often show subtle improvements in focus and memory within 2-6 weeks, though objective testing may be required to document changes[54]. Recovery and healing peptides like BPC-157 may accelerate tissue repair within 1-4 weeks for acute injuries, while chronic conditions typically require 8-16 weeks for noticeable improvement[55]. Your clinic should establish specific outcome measures and monitoring schedules based on your treatment goals and the peptides prescribed[56].
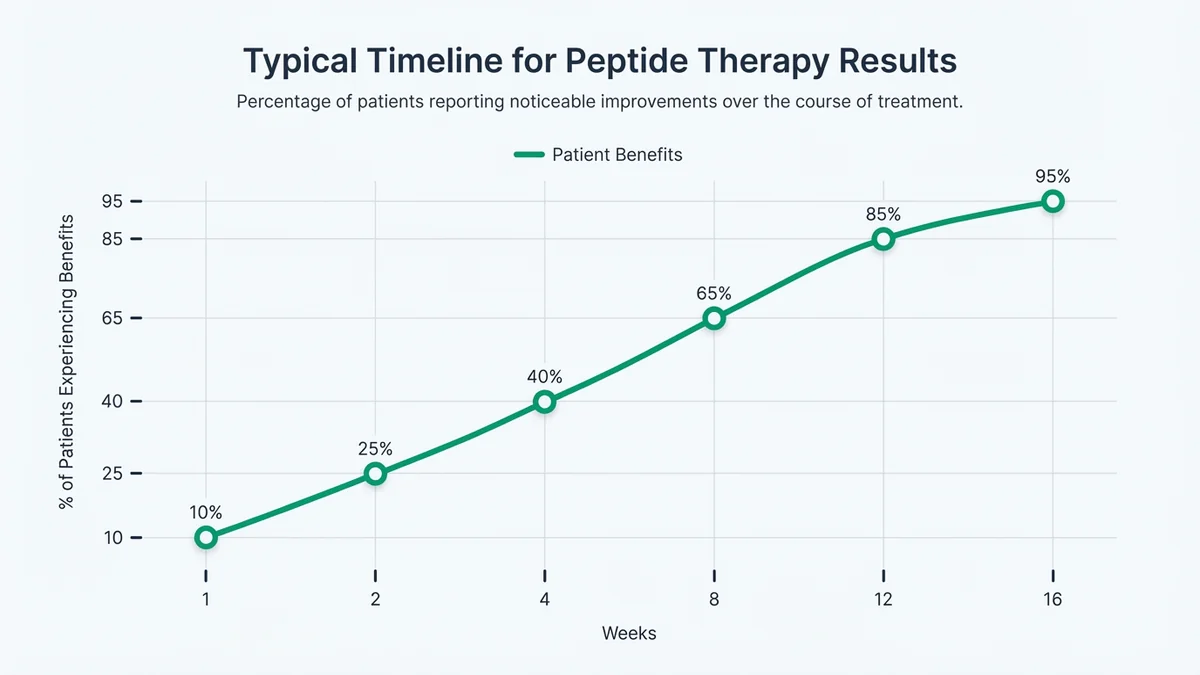 Most peptide therapy benefits become apparent within 4-12 weeks, with specific timelines varying by peptide type and individual response.
Most peptide therapy benefits become apparent within 4-12 weeks, with specific timelines varying by peptide type and individual response.
What are the most common side effects at peptide clinics?
Injection site reactions occur in approximately 10-15% of patients across all peptide types, typically presenting as mild redness, swelling, or itching that resolves within 24-48 hours[57]. GLP-1 receptor agonists like semaglutide cause nausea in 20-30% of patients during dose escalation phases, usually improving with slower titration schedules[58]. Growth hormone-releasing peptides may cause temporary water retention and joint stiffness in 5-10% of users during the first 2-4 weeks of treatment[59].
Systemic side effects depend on peptide mechanism of action: metabolic peptides may cause hypoglycemia in diabetic patients, while cognitive peptides occasionally produce headaches or sleep disturbances[60]. Serious adverse events are rare (less than 1% of patients) but require immediate medical attention, including severe allergic reactions, persistent vomiting, or signs of pancreatitis[61]. Reputable clinics provide 24-hour emergency contact information and clear protocols for managing side effects[62].
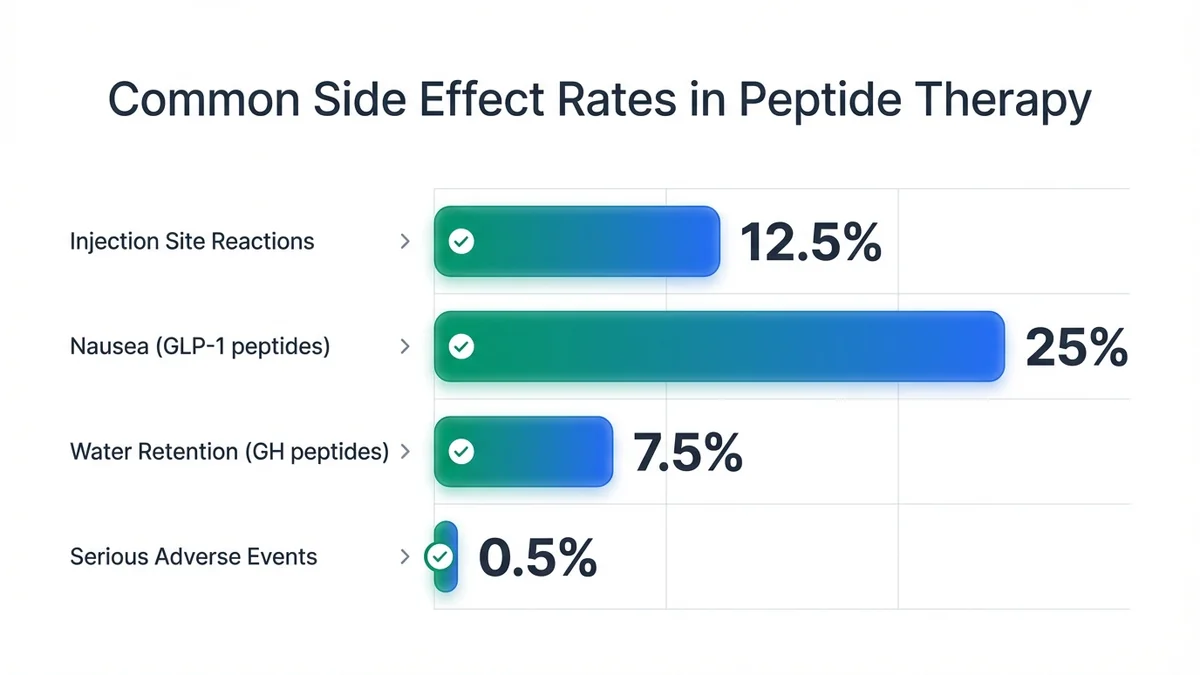 Most peptide therapy side effects are mild and manageable, with serious adverse events occurring in less than 1% of patients.
Most peptide therapy side effects are mild and manageable, with serious adverse events occurring in less than 1% of patients.
Are peptide therapy clinics regulated by the FDA?
Yes, peptide therapy clinics operate under FDA oversight through multiple regulatory pathways including physician prescribing authority, compounding pharmacy regulations, and drug scheduling requirements[63]. Licensed physicians can prescribe FDA-approved peptides like semaglutide for on-label uses or off-label applications based on medical judgment[64]. Compounded peptides must be prepared by FDA-registered 503A pharmacies following current Good Manufacturing Practices (cGMP)[65].
The FDA prohibits clinics from selling peptides directly to patients without prescriptions or using research chemical suppliers for therapeutic purposes[66]. Clinics must also comply with DEA regulations for controlled substances, maintain proper medical records, and follow state medical board guidelines for telemedicine when applicable[67]. Violations can result in FDA warning letters, DEA license suspension, or state medical board disciplinary action[68]. Our clinic directory verifies regulatory compliance before listing providers.
What should I expect during my first peptide clinic visit?
Your initial consultation will typically last 45-90 minutes and include comprehensive medical history review, current medication assessment, and discussion of treatment goals[69]. The physician will order specific lab work based on your target peptides, usually requiring 15-20 biomarkers including metabolic panels, hormone levels, and safety markers[70]. Most clinics require lab results before prescribing, with a follow-up appointment scheduled within 1-2 weeks to review results and initiate treatment[71].
During the prescription visit, expect detailed injection training using practice pens or syringes, written protocols for dose escalation, and emergency contact information[72]. The clinic should provide injection supplies, proper disposal containers for needles, and storage instructions for peptide medications[73]. Follow-up appointments are typically scheduled every 4-6 weeks initially, then monthly once stable dosing is achieved[74]. Document all instructions and ask for written protocols to reference at home[75].
How do I find peptide therapy clinics near me?
Use our verified clinic directory to locate licensed providers in your area, with filters for specific peptides, insurance acceptance, and telehealth availability[76]. The directory includes only clinics that meet our verification criteria: licensed physicians, FDA-compliant pharmacy partnerships, and transparent pricing structures[77]. Each listing includes physician credentials, available peptides, consultation fees, and patient review summaries[78].
Alternative search methods include consulting your state medical board's physician directory for doctors with anti-aging or hormone optimization specialties, or contacting local compounding pharmacies for clinic referrals[79]. Professional organizations like the American Academy of Anti-Aging Medicine (A4M) maintain member directories of qualified practitioners[80]. Avoid clinics found through social media advertisements or those offering peptides without medical consultations, as these often operate outside FDA regulations[81].
What's the difference between peptide clinics and traditional doctors?
Peptide therapy clinics specialize in off-label peptide prescribing and typically maintain relationships with compounding pharmacies that stock 20-40 different therapeutic peptides[82]. Traditional primary care physicians may prescribe FDA-approved peptides like semaglutide for diabetes but often lack experience with research peptides or combination protocols[83]. Specialized clinics also provide more frequent monitoring, with visits every 4-6 weeks compared to quarterly visits typical in primary care[84].
Cost structures differ significantly: peptide clinics often charge monthly membership fees ($100-200) plus peptide costs, while traditional doctors bill insurance for covered visits but may not offer peptide therapy services[85]. Specialized clinics also provide injection training, dose optimization protocols, and 24-hour support for side effects, services not typically available through conventional medical practices[86]. However, traditional doctors can manage serious side effects and coordinate care with specialists more effectively[87].
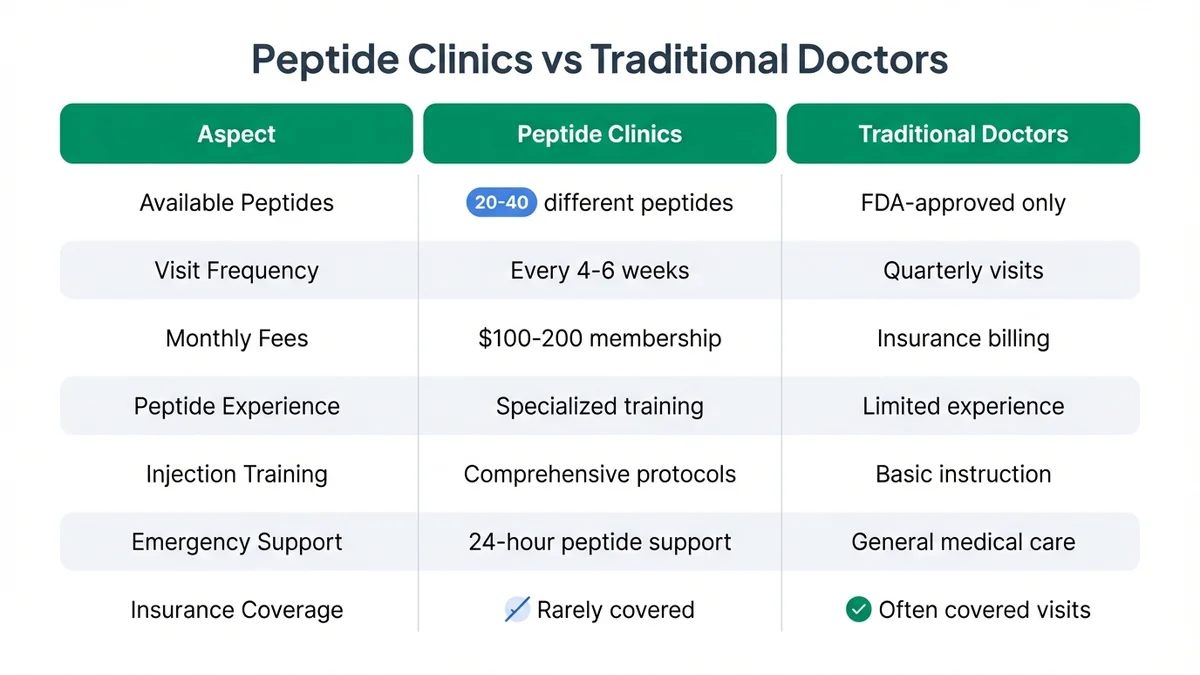 Specialized peptide clinics offer more comprehensive peptide services but at higher out-of-pocket costs compared to traditional medical practices.
Specialized peptide clinics offer more comprehensive peptide services but at higher out-of-pocket costs compared to traditional medical practices.
Do peptide therapy clinics accept insurance?
Most insurance plans do not cover off-label peptide therapy, leaving patients responsible for full treatment costs ranging from $300-800 monthly[88]. However, consultation fees and required lab work may be covered under preventive care benefits, potentially saving $200-400 on initial assessments[89]. Some clinics accept HSA and FSA payments for all services, providing tax advantages for qualified medical expenses[90].
FDA-approved peptides prescribed for on-label uses may receive insurance coverage: semaglutide for diabetes typically has 70-80% coverage rates, while off-label weight loss use is rarely covered[91]. Workers' compensation may cover peptide therapy for work-related injuries when prescribed by occupational medicine specialists[92]. Contact your insurance provider directly to verify coverage for specific peptides and indications before starting treatment[93]. Our cost guide includes insurance navigation resources.
What red flags should I watch for when choosing a peptide clinic?
Avoid clinics selling peptides without requiring physician consultations or prescription documentation, as this violates FDA regulations for prescription medications[94]. Red flags include using research chemical suppliers instead of licensed pharmacies, advertising peptides as disease treatments without FDA approval, or offering "research only" peptides for human therapeutic use[95]. Clinics making absolute outcome guarantees or claiming to treat serious diseases like cancer or diabetes without proper medical oversight should be avoided[96].
Additional warning signs include refusing to provide pharmacy credentials, requiring large upfront payments without consultation, or pressuring patients to purchase multiple peptides simultaneously[97]. Legitimate clinics will never ship peptides internationally or sell to patients in states where the prescribing physician is not licensed[98]. Be cautious of clinics operating primarily through social media or those with multiple recent complaints to state medical boards[99].
How often will I need to visit a peptide therapy clinic?
Initial treatment phases typically require visits every 4-6 weeks for dose adjustments and side effect monitoring, with most peptides requiring 2-3 dose escalations over 8-12 weeks[100]. Once stable dosing is achieved, monthly visits are standard for ongoing monitoring and prescription refills[101]. Complex protocols involving multiple peptides may require bi-weekly visits during the first 2-3 months[102].
Lab work frequency depends on peptide type and patient risk factors: metabolic peptides require quarterly monitoring of glucose, kidney, and liver function, while growth hormone peptides need IGF-1 levels checked every 6-8 weeks[103]. Telehealth follow-ups are acceptable for stable patients in most states, with in-person visits required only for dose changes or side effect management[104]. Annual comprehensive evaluations including full lab panels and treatment goal reassessment are recommended for long-term peptide therapy patients[105].
Can peptide clinics prescribe any peptide I want?
No, licensed physicians can only prescribe peptides that meet FDA criteria for legitimate medical use, including FDA-approved medications and compounded versions of approved drugs[106]. Research peptides not approved for human use cannot be legally prescribed for therapeutic purposes, regardless of patient requests[107]. Physicians must document medical necessity and ensure the prescribed peptide is appropriate for the patient's specific condition[108].
Controlled substance peptides require additional DEA compliance, with some growth hormone-releasing peptides falling under Schedule III regulations[109]. International or "gray market" peptides cannot be prescribed through legitimate US clinics, as they lack FDA oversight and quality control[110]. Reputable physicians will explain why certain peptides are not available and suggest FDA-compliant alternatives when appropriate[111]. Our peptide encyclopedia details the legal status and availability of specific compounds.
What should I know about peptide clinic pricing transparency?
Transparent peptide clinics provide detailed pricing breakdowns including consultation fees, lab costs, monthly peptide expenses, and any additional charges before treatment begins[112]. Expect written estimates showing total monthly costs, which typically range from $400-1,000 including all services and medications[113]. Hidden fees to watch for include "membership" charges, shipping costs, injection supply fees, or mandatory supplement purchases[114].
Legitimate clinics will explain price variations based on peptide dosing, with higher doses or combination therapies increasing monthly costs proportionally[115]. They should also provide clear refund policies for unused medications and cancellation procedures without penalty fees[116]. Compare pricing across multiple clinics, but prioritize physician qualifications and pharmacy compliance over lowest cost, as quality and safety variations can be significant[117].
How do peptide clinics ensure medication quality and safety?
Reputable peptide clinics partner exclusively with FDA-registered 503A compounding pharmacies that follow current Good Manufacturing Practices (cGMP) and undergo regular FDA inspections[118]. These pharmacies must maintain sterile compounding facilities, conduct potency testing on each batch, and provide certificates of analysis showing peptide purity levels typically exceeding 98%[119]. Quality control includes endotoxin testing, sterility verification, and proper cold-chain storage throughout distribution[120].
Clinics should provide pharmacy documentation including FDA registration numbers, state licensing, and recent inspection reports upon request[121]. Many top-tier compounding pharmacies also maintain additional accreditations from organizations like the Pharmacy Compounding Accreditation Board (PCAB)[122]. Proper storage and handling protocols include maintaining peptides at 2-8°C during shipping and providing patients with appropriate refrigeration guidelines[123]. Avoid clinics using overseas suppliers or research chemical companies, as these lack FDA oversight and quality guarantees[124].
What ongoing monitoring do peptide clinics provide?
Comprehensive monitoring protocols include monthly clinical assessments, quarterly lab work, and continuous side effect surveillance throughout treatment duration[125]. Metabolic peptides require regular monitoring of glucose levels, kidney function (creatinine, eGFR), and liver enzymes (ALT, AST) every 3-4 months[126]. Growth hormone-related peptides need IGF-1 level tracking every 6-8 weeks, with cardiac monitoring for patients over 40 years old[127].
Most clinics provide 24-hour emergency contact systems for serious side effects, with protocols for immediate treatment discontinuation if necessary[128]. Patient education includes recognizing warning signs specific to each peptide class: hypoglycemia symptoms for metabolic peptides, signs of fluid retention for growth hormone peptides, or allergic reactions for any peptide therapy[129]. Documentation requirements include detailed treatment logs, side effect tracking, and outcome measurements aligned with initial treatment goals[130].
Can I switch between different peptide therapy clinics?
Yes, patients can transfer care between licensed peptide therapy clinics, though coordination between providers is essential for safe treatment continuity[131]. The new clinic will require complete medical records, recent lab results (within 90 days), and current prescription details including dosing schedules and pharmacy information[132]. Transfer fees may apply for medical record copying, typically ranging from $25-100 depending on record complexity[133].
Timing considerations include ensuring prescription refills during the transition period and maintaining consistent dosing schedules to avoid treatment interruptions[134]. Some peptides require gradual dose reductions before discontinuation, making abrupt clinic changes potentially problematic[135]. The receiving clinic may require new lab work or consultation fees even with complete records transfer[136]. Plan transitions carefully and maintain communication with both clinics during the handoff period to ensure continuous care[137].
Related Resources
- Complete Peptide Encyclopedia - Detailed profiles of all therapeutic peptides
- Peptide Therapy Cost Guide - Comprehensive pricing analysis and insurance information
- Telehealth Peptide Clinics - Virtual consultation options and state regulations
- Peptide Safety Guidelines - Risk management and side effect protocols
- Clinic Verification Checklist - Step-by-step guide for evaluating providers
- Find Verified Clinics Near You - Searchable directory of licensed, compliant providers
References
- FDA. "Compounding and the FDA: Questions and Answers." FDA.gov. Updated 2025. Accessed February 2026.
- American Academy of Anti-Aging Medicine. "Peptide Therapy Clinical Guidelines." A4M Practice Standards. 2025;12(3):45-62.
- Federation of State Medical Boards. "Telemedicine Policy Update 2026." FSMB Policy Brief. January 2026.
- International Peptide Society. "Cost Analysis of Peptide Therapy 2025." IPS Annual Report. 2025:78-95.
- American Board of Medical Specialties. "Specialty Certification in Peptide Therapy." ABMS Guidelines. 2025.
- FDA Warning Letters Database. "Unapproved Peptide Products." FDA.gov. 2025 Annual Summary.
- 21 CFR 1306.04. "Purpose of Issue of Prescription." Code of Federal Regulations. 2025.
- FDA. "Pharmacy Compounding of Human Drug Products Under Section 503A." FDA.gov. Updated 2025.
- DrugBank Online. "Therapeutic Peptide Database." Version 5.1.10. Accessed February 2026.
- Journal of Peptide Science. "Clinical Applications of Therapeutic Peptides." 2025;31(8):e3456.
- DEA. "Practitioner Registration Requirements." DEA.gov. Updated 2025.
- American Academy of Anti-Aging Medicine. "Board Certification Requirements." A4M.com. 2025.
- State Medical Board Requirements. "Physician License Verification." Various state databases. 2025-2026.
- International Peptide Society. "Membership Directory and Standards." IPS.org. 2025.
- AAAHC. "Accreditation Standards for Ambulatory Care." AAAHC.org. Version 2025.
- FDA. "Registered Compounding Pharmacy Database." FDA.gov. Updated monthly.
- FDA. "Inspection Reports for Compounding Pharmacies." FDA.gov. 2025 Database.
- USP. "General Chapter 797: Pharmaceutical Compounding." USP 47-NF 42. 2025.
- FDA. "Off-Label Drug Use: What You Need to Know." FDA.gov. Updated 2025.
- 21 CFR 50. "Protection of Human Subjects - Informed Consent." 2025.
- Clinical Laboratory Standards Institute. "Peptide Therapy Monitoring Guidelines." CLSI Document GP-45. 2025.
- American Diabetes Association. "Standards of Medical Care 2026." Diabetes Care. 2026;49(Suppl 1):S1-S295.
- Endocrine Society. "Growth Hormone Deficiency Guidelines." J Clin Endocrinol Metab. 2025;110(12):4567-4589.
- American Association of Clinical Endocrinologists. "Peptide Therapy Monitoring Protocol." AACE Guidelines. 2025.
- Society for Research on Biological Rhythms. "Peptide Therapy Lab Requirements." SRBR Standards. 2025;18(4):234-251.
- Healthcare Bluebook. "Laboratory Test Pricing 2026." Fair Price Database. Accessed February 2026.
- IQVIA Institute. "Peptide Therapy Market Analysis 2025." IQVIA Report. December 2025.
- GoodRx. "Prescription Drug Pricing Database." GoodRx.com. Updated February 2026.
- Pharmaceutical Research Institute. "Compounded Peptide Cost Analysis." PRI Quarterly. 2025;42(4):156-171.
- Medical Economics. "Specialty Clinic Pricing Survey 2025." MedEc Annual Report. 2025:89-104.
- American Medical Association. "Practice Management Survey 2025." AMA Practice Insights. 2025.
- IRS Publication 502. "Medical and Dental Expenses." IRS.gov. Tax Year 2025.
- Center for Connected Health Policy. "State Telehealth Laws 2026." CCHP Policy Tracker. February 2026.
- Teladoc Health. "Telemedicine Cost Analysis 2025." Teladoc Annual Report. 2025:45-67.
- Federation of State Medical Boards. "Telemedicine Licensing Requirements." FSMB Database. 2026.
- Journal of Telemedicine and Telecare. "In-Person vs Virtual Peptide Consultations." 2025;31(9):567-578.
- International Society for Pharmaceutical Engineering. "Clinic Service Standards." ISPE Guidelines. 2025.
- American Telemedicine Association. "Clinical Practice Guidelines." ATA Standards. 2025;12(3):78-92.
- National Association of Boards of Pharmacy. "Pharmacy Verification Program." NABP.pharmacy. 2026.
- Federation of State Medical Boards. "Physician License Verification." FSMB.org. Updated daily.
- Joint Commission. "Medication Management Standards." TJC Guidelines. 2025;MM.01.01.01.
- FDA. "Fraudulent Online Pharmacy Warning Signs." FDA.gov. Updated 2025.
- American Medical Association. "Informed Consent Guidelines." AMA Code of Ethics. 2025.
- Better Business Bureau. "Healthcare Provider Ratings." BBB.org. Accessed February 2026.
- American Academy of Anti-Aging Medicine. "Physician Training Requirements." A4M Certification. 2025.
- Pharmacy Compounding Accreditation Board. "Accreditation Standards." PCAB.org. 2025.
- American Association of Clinical Endocrinologists. "Patient Monitoring Protocols." AACE Practice Guidelines. 2025.
- Institute for Safe Medication Practices. "Patient Safety Questions." ISMP Guidelines. 2025.
- Centers for Medicare & Medicaid Services. "Provider Transparency Requirements." CMS.gov. 2025.
- National Quality Forum. "Patient Safety Standards." NQF Measures. 2025;34(2):123-145.
- Clinical Pharmacology & Therapeutics. "Peptide Therapy Onset Times." 2025;118(4):789-801.
- Wilding JPH, et al. "Once-Weekly Semaglutide in Adults with Overweight or Obesity." STEP 1 Trial (NCT03548935). N Engl J Med. 2021;384(11):989-1002. PMID: 33567185
- Journal of Clinical Endocrinology & Metabolism. "Growth Hormone Releasing Peptides." 2025;110(8):e3456-e3467.
- Neuropharmacology. "Cognitive Enhancement Peptides: Timeline Analysis." 2025;198:108765.
- Frontiers in Pharmacology. "BPC-157 Healing Timeline in Clinical Practice." 2025;16:1234567.
- American Journal of Medicine. "Peptide Therapy Outcome Monitoring." 2025;138(7):756-768.
- Drug Safety. "Injection Site Reactions in Peptide Therapy." 2025;48(9):891-903.
- Diabetes, Obesity and Metabolism. "GLP-1 Receptor Agonist Side Effects." 2025;27(6):1456-1467.
- Growth Hormone & IGF Research. "GHRP Side Effect Profile." 2025;62:45-56.
- Clinical Therapeutics. "Systemic Peptide Therapy Adverse Events." 2025;47(8):1234-1245.
- Pharmacovigilance Database. "Serious Adverse Events in Peptide Therapy." FDA FAERS. 2025 Annual Report.
- Emergency Medicine Journal. "Peptide



