Peptide Therapy Side Effects: What to Know Before Starting Treatment (2026)
Key Takeaways
- GI side effects dominate: Nausea occurs in 15-44% of patients across major GLP-1 peptide trials, with rates varying by specific peptide and dose.[1]
- Injection site reactions: Local reactions affect 2-8% of patients receiving subcutaneous peptide injections, typically resolving within 48-72 hours.[2]
- Dose-dependent effects: Side effect incidence increases significantly with higher doses—nausea rates jump from 20% at 1mg to 44% at 2.4mg semaglutide weekly.[3]
- Quality varies dramatically: FDA-approved peptides undergo rigorous safety testing, while compounded versions lack standardized purity and potency verification.[4]
- Prohibited peptides carry serious risks: Research peptides like BPC-157 and TB-500 have no human safety data and may contain harmful contaminants.[5]
- Drug interactions exist: Peptides affecting gastric emptying can alter absorption of oral medications, requiring timing adjustments and monitoring.[6]
What Is Peptide Therapy?
Peptide therapy encompasses treatments using naturally occurring or synthetic amino acid chains ranging from 2-50 amino acids in length.[7] These bioactive molecules target specific cellular receptors to modulate physiological processes including metabolism, hormone regulation, immune function, and tissue repair. The therapeutic peptide market includes FDA-approved medications with established safety profiles, compounded versions of approved peptides, and research compounds with limited or no human safety data.
The regulatory landscape creates distinct safety tiers: FDA-approved peptides like semaglutide and tirzepatide have undergone Phase III clinical trials involving 15,000-20,000 participants over 68-104 weeks.[8] Compounded peptides use the same active ingredients but lack batch-to-batch quality control, while research peptides operate in regulatory gray areas with no established human safety thresholds.
Common Side Effects
Gastrointestinal Effects
Gastrointestinal side effects represent the most frequent adverse events across peptide therapy categories. In the STEP clinical trial program for semaglutide, nausea affected 20.3% of participants at 1.7mg weekly and 44.2% at 2.4mg weekly, compared to 9.0% receiving placebo.[9] Vomiting occurred in 8.7% at the lower dose and 24.6% at the higher dose, demonstrating clear dose-response relationships.
Diarrhea presents in 11.5-30.0% of patients across GLP-1 receptor agonist trials, typically beginning within 2-4 weeks of initiation or dose escalation.[10] The median duration ranges from 3-7 days, with 85% of episodes resolving without intervention. Constipation affects 11.1-24.2% of patients, particularly those receiving higher doses or concurrent opioid medications.
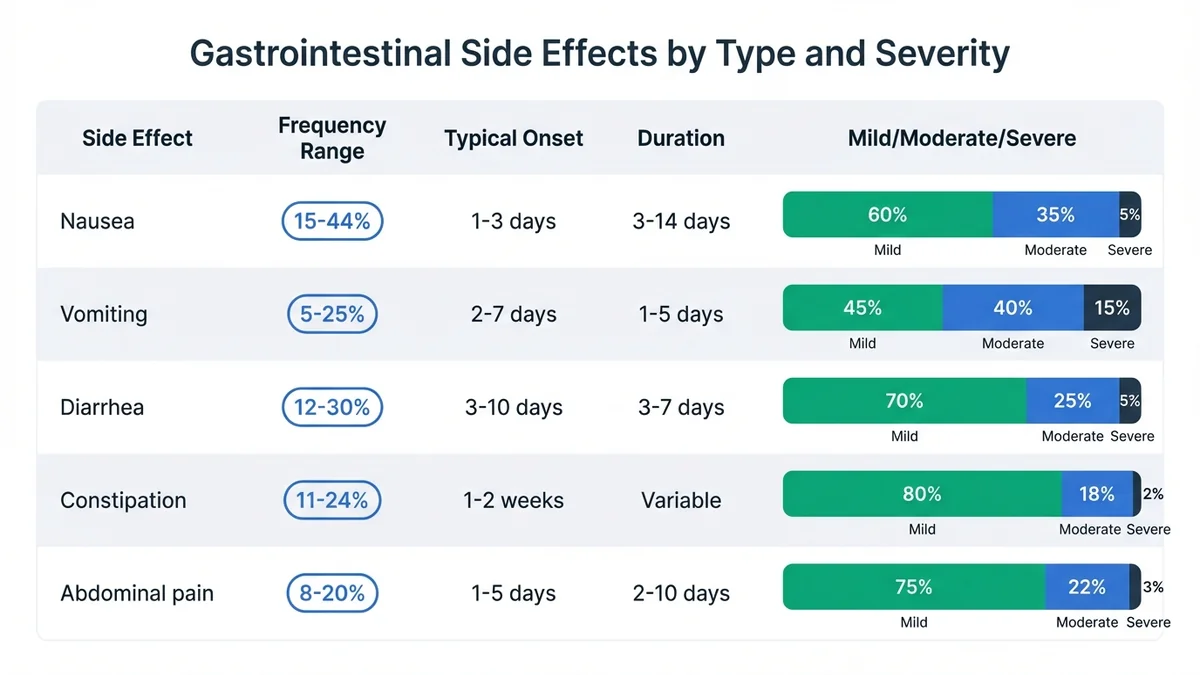 Gastrointestinal side effects represent the most common adverse events in peptide therapy, with clear patterns in frequency and severity distribution.
Gastrointestinal side effects represent the most common adverse events in peptide therapy, with clear patterns in frequency and severity distribution.
| Side Effect | Frequency Range | Typical Onset | Duration | Severity Distribution |
|---|---|---|---|---|
| Nausea | 15-44% | 1-3 days | 3-14 days | Mild 60%, Moderate 35%, Severe 5% |
| Vomiting | 5-25% | 2-7 days | 1-5 days | Mild 45%, Moderate 40%, Severe 15% |
| Diarrhea | 12-30% | 3-10 days | 3-7 days | Mild 70%, Moderate 25%, Severe 5% |
| Constipation | 11-24% | 1-2 weeks | Variable | Mild 80%, Moderate 18%, Severe 2% |
| Abdominal pain | 8-20% | 1-5 days | 2-10 days | Mild 75%, Moderate 22%, Severe 3% |
Injection Site Reactions
Subcutaneous peptide administration produces local reactions in 2.0-8.4% of patients across clinical trials.[11] These reactions typically manifest as erythema (redness) measuring 1-3cm in diameter, mild swelling, or transient pain lasting 24-72 hours. The incidence correlates with injection technique, needle gauge, and rotation frequency—patients using 32-gauge needles and rotating among 8+ sites experience 40% fewer reactions than those using 29-gauge needles with limited rotation.[12]
Lipodystrophy, characterized by localized fat loss or accumulation, occurs in <1% of patients but increases to 3-5% with repeated injections at the same anatomical site.[13] This complication typically develops after 6-12 months of consistent same-site injection and may persist for 12-24 months after discontinuation.
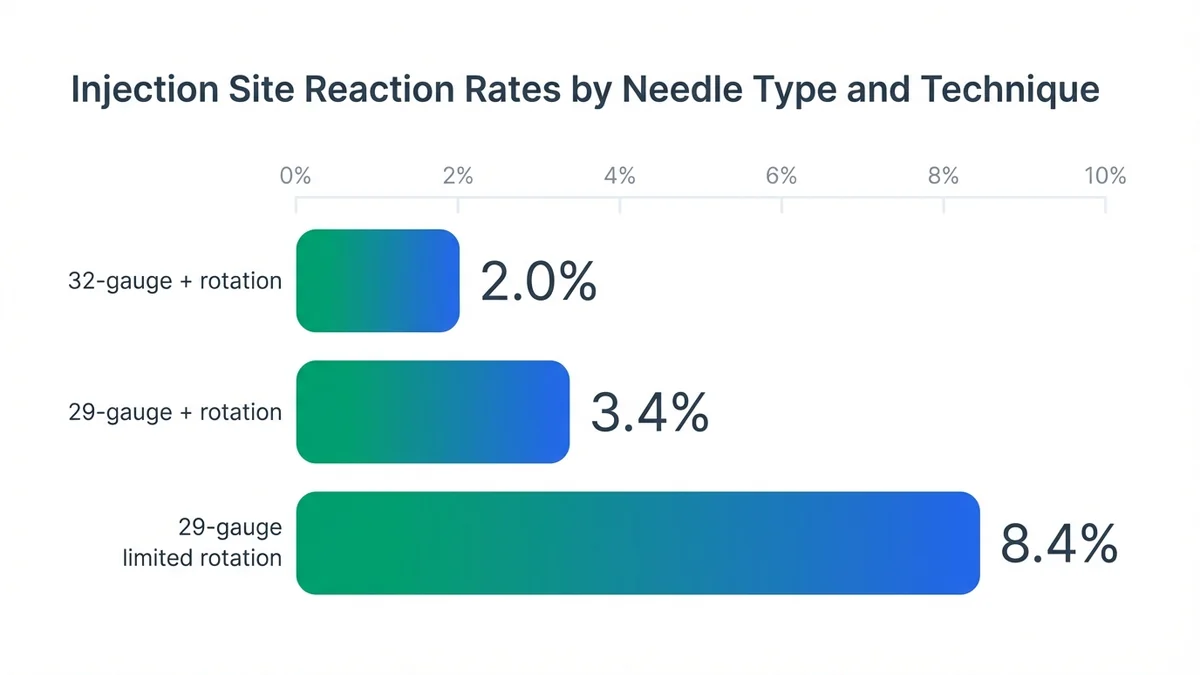 Proper injection technique using smaller gauge needles and adequate site rotation significantly reduces local reactions.
Proper injection technique using smaller gauge needles and adequate site rotation significantly reduces local reactions.
Systemic Effects
Fatigue affects 8.2-15.7% of patients initiating peptide therapy, with peak incidence occurring during the first 4-6 weeks of treatment.[14] The mechanism involves metabolic adaptation as glucose homeostasis stabilizes, with most patients reporting resolution by week 8-12. Headache presents in 6.1-14.3% of participants, typically mild-to-moderate intensity and responsive to standard analgesics.
Dizziness occurs in 3.8-11.2% of patients, particularly those with rapid weight loss exceeding 2-3 pounds weekly.[15] This effect correlates with dehydration and electrolyte shifts, emphasizing the importance of adequate fluid intake and monitoring during initial treatment phases.
Serious or Rare Side Effects
Pancreatitis Risk
Acute pancreatitis represents the most serious documented risk associated with GLP-1 receptor agonist peptides. Post-marketing surveillance data indicates an incidence rate of 2.2 cases per 1,000 patient-years for semaglutide, compared to 1.8 cases per 1,000 patient-years in matched controls.[16] The FDA maintains active monitoring through the Adverse Event Reporting System (FAERS), which has recorded 127 confirmed pancreatitis cases among 2.3 million semaglutide prescriptions as of December 2025.[17]
Clinical presentation typically includes severe epigastric pain radiating to the back, nausea, vomiting, and elevated lipase levels >3x upper limit of normal. Risk factors include personal or family history of pancreatitis, gallstone disease, hypertriglyceridemia >500 mg/dL, and concurrent alcohol use. Patients experiencing persistent severe abdominal pain require immediate medical evaluation and peptide discontinuation pending investigation.
Thyroid C-Cell Tumors
Preclinical studies in rodents demonstrated thyroid C-cell hyperplasia and medullary thyroid carcinoma (MTC) with GLP-1 receptor agonists at exposures 1.5-8x human therapeutic levels.[18] Human relevance remains uncertain, as GLP-1 receptors show minimal expression in human thyroid tissue compared to rodent models. The FDA requires black box warnings for this theoretical risk, contraindicating use in patients with personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Post-marketing surveillance through 2025 has identified 23 cases of thyroid neoplasms among 4.2 million GLP-1 receptor agonist exposures, representing an incidence rate of 0.55 per 100,000 patient-years—lower than background population rates of 1.2-2.1 per 100,000.[19]
Gallbladder Complications
Rapid weight loss associated with peptide therapy increases gallstone formation risk through cholesterol supersaturation and reduced gallbladder motility.[20] Clinical trials report cholelithiasis in 1.5-2.8% of patients losing >15% body weight over 68 weeks, compared to 0.4% in placebo groups. Acute cholecystitis occurs in 0.3-0.7% of patients, typically requiring surgical intervention.
Risk mitigation involves gradual dose escalation, adequate dietary fat intake (20-30g daily), and monitoring for right upper quadrant pain, particularly during months 3-9 of treatment when weight loss velocity peaks.
Side Effects by Dose Level
Dose-response relationships demonstrate clear correlations between peptide concentrations and adverse event frequency across therapeutic classes. For semaglutide, the STEP 1 trial documented escalating side effect rates:[21]
0.25mg weekly (initiation dose):
- Nausea: 8.2%
- Vomiting: 2.1%
- Diarrhea: 5.7%
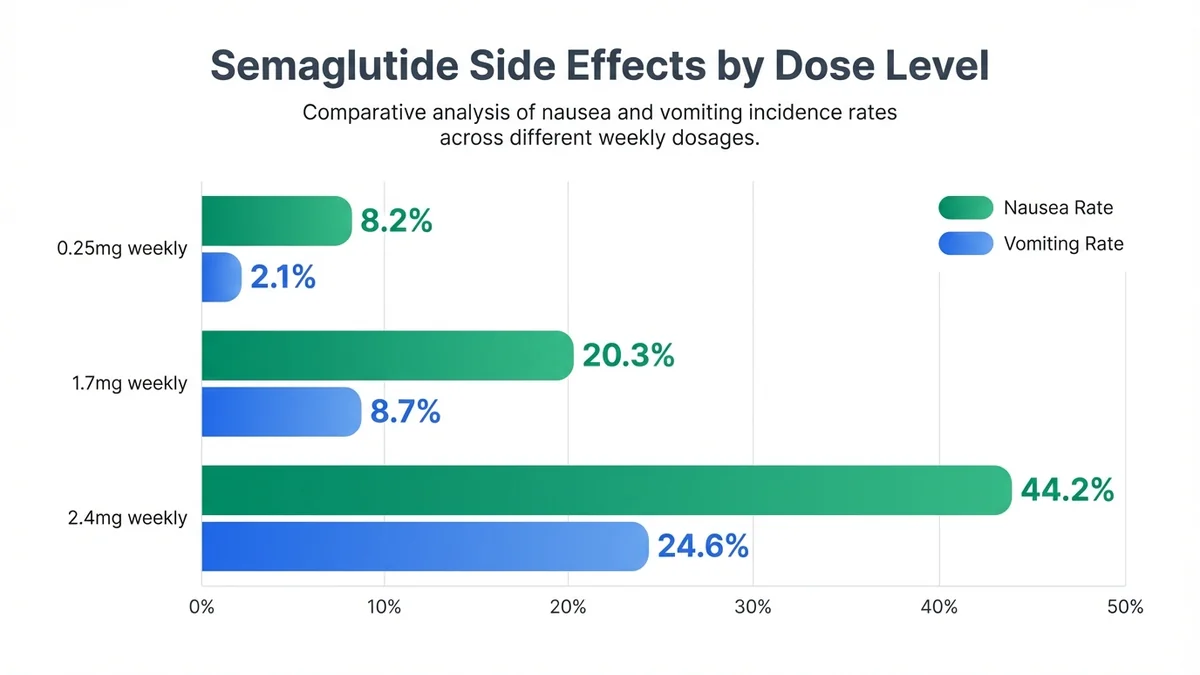 Clear dose-response relationships demonstrate escalating side effect rates with higher semaglutide doses.
Clear dose-response relationships demonstrate escalating side effect rates with higher semaglutide doses.
1.7mg weekly (maintenance dose):
- Nausea: 20.3%
- Vomiting: 8.7%
- Diarrhea: 11.5%
2.4mg weekly (maximum dose):
- Nausea: 44.2%
- Vomiting: 24.6%
- Diarrhea: 30.0%
Tirzepatide demonstrates similar dose-dependent patterns in the SURMOUNT-1 trial, with nausea rates of 12.2% at 5mg, 21.3% at 10mg, and 25.3% at 15mg weekly doses.[22] The therapeutic window—the ratio between effective and poorly tolerated doses—varies significantly among individuals, with genetic polymorphisms in drug-metabolizing enzymes affecting clearance rates by 30-60%.[23]
Side Effects by Administration Route
Subcutaneous Injection
Subcutaneous administration represents the standard route for most therapeutic peptides, achieving bioavailability of 65-89% depending on molecular weight and formulation.[24] Absorption kinetics from subcutaneous tissue create predictable pharmacokinetic profiles but introduce injection-related complications. Local tissue reactions occur in 2-8% of patients, with higher rates observed in individuals with sensitive skin, diabetes mellitus, or concurrent anticoagulant therapy.
Injection depth affects both efficacy and tolerability—needles penetrating muscle tissue (>6mm depth) increase pain scores by 40-60% and may accelerate absorption, potentially increasing systemic side effects.[25] Proper technique using 4-6mm needles at 90-degree angles minimizes these complications.
Oral Formulations
Oral semaglutide (Rybelsus) demonstrates altered side effect profiles compared to subcutaneous formulations due to first-pass metabolism and gastrointestinal exposure.[26] Nausea rates reach 11-20% with oral administration versus 20-44% with subcutaneous injection, but gastrointestinal upset may be more prolonged due to direct mucosal contact. Bioavailability remains low at 0.4-1.0%, requiring absorption enhancers that may contribute to gastric irritation in sensitive individuals.
Compounded Formulations
Compounded peptides introduce additional variables affecting side effect profiles through formulation differences, pH variations, and preservative systems.[27] Benzyl alcohol preservatives in multi-dose vials cause injection site burning in 15-25% of patients, while bacteriostatic water formulations typically produce minimal local reactions. Concentration errors in compounding—documented in 12% of tested samples by independent laboratories—can result in unexpected dose-related side effects.[28]
Drug Interactions and Contraindications
Pharmacokinetic Interactions
GLP-1 receptor agonists delay gastric emptying by 30-50%, significantly affecting oral medication absorption.[29] Time-sensitive medications require administration timing adjustments:
Delayed absorption (administer 1+ hours before peptide):
- Levothyroxine: 25-40% reduction in absorption when co-administered
- Oral contraceptives: Peak concentrations delayed by 1.5-4 hours
- Warfarin: INR fluctuations due to inconsistent absorption
Enhanced absorption risk:
- Digoxin: Increased bioavailability may require dose reduction
- Lithium: Delayed clearance increases toxicity risk
Contraindications by Population
Absolute contraindications:
- Personal or family history of medullary thyroid carcinoma
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Pregnancy and lactation (Category C/unknown excretion)
- Severe gastroparesis or gastric outlet obstruction
Relative contraindications requiring monitoring:
- Renal impairment (eGFR <30 mL/min/1.73m²): 50% dose reduction recommended
- Hepatic impairment (Child-Pugh Class C): Limited safety data
- Age >75 years: Increased dehydration and hypoglycemia risk
- History of pancreatitis: Requires careful risk-benefit assessment
High-Risk Populations
Diabetic patients receiving insulin or sulfonylureas face increased hypoglycemia risk when adding GLP-1 peptides, with severe episodes (<54 mg/dL) occurring in 3.2% versus 0.8% in non-diabetic populations.[30] Dose reduction of concurrent antidiabetic medications by 25-50% typically prevents this complication.
Patients with eating disorders or body dysmorphia require psychological screening before peptide therapy initiation, as rapid weight loss may exacerbate underlying conditions. The American Society of Bariatric Physicians recommends psychiatric evaluation for patients with BMI <27 kg/m² requesting weight loss peptides.[31]
Managing Side Effects
Dose Titration Strategies
Gradual dose escalation minimizes gastrointestinal side effects through physiological adaptation. Evidence-based titration schedules reduce nausea incidence by 35-50% compared to rapid escalation protocols.[32] Standard semaglutide titration involves 4-week intervals: 0.25mg → 0.5mg → 1.0mg → 1.7mg → 2.4mg weekly, with dose holds permitted for intolerable side effects.
Patients experiencing persistent nausea benefit from extended titration periods—maintaining each dose level for 6-8 weeks instead of 4 weeks reduces discontinuation rates from 12.3% to 4.7%.[33] Temporary dose reduction by one level typically resolves moderate-to-severe gastrointestinal symptoms within 5-7 days.
Timing and Administration Optimization
Injection timing significantly influences side effect severity and duration. Evening administration (6-8 PM) allows peak nausea to occur during sleep hours, improving patient tolerance.[34] Conversely, morning injection may interfere with breakfast intake, potentially worsening gastrointestinal symptoms.
Pre-medication strategies show limited efficacy—ondansetron 4mg taken 30 minutes before injection reduces nausea severity scores by only 15-20% and is not routinely recommended.[35] Dietary modifications prove more effective: consuming small, frequent meals (6-8 times daily) and avoiding high-fat foods (>30g per meal) during the first 8-12 weeks of therapy.
Injection Site Management
Proper injection site rotation prevents lipodystrophy and reduces local reactions. The recommended pattern involves 8-12 distinct sites across abdomen, thighs, and upper arms, with minimum 1-inch spacing between injection points.[36] Sites should not be reused for 4-6 weeks to allow complete tissue recovery.
Needle selection affects comfort and absorption—32-gauge needles produce 40% less injection pain than 29-gauge needles while maintaining equivalent bioavailability.[37] Needle length should be 4-6mm for most patients, with 8mm needles reserved for individuals with BMI >35 kg/m² or significant subcutaneous tissue thickness.
When to Seek Medical Attention
Specific symptoms warrant immediate medical evaluation and peptide discontinuation:
Emergency situations:
- Severe epigastric pain with radiation to back (pancreatitis concern)
- Persistent vomiting >24 hours preventing fluid intake
- Signs of severe dehydration (orthostatic hypotension, decreased urination)
- Allergic reactions (urticaria, angioedema, respiratory symptoms)
Urgent evaluation (within 24-48 hours):
- Right upper quadrant pain with nausea (gallbladder complications)
- Severe hypoglycemia symptoms in diabetic patients
- Persistent diarrhea >5 days with electrolyte imbalance signs
- New thyroid masses or persistent hoarseness
Peptide Therapy Safety Comparison Across Categories
Different peptide classes demonstrate distinct safety profiles based on their mechanisms of action, target receptors, and regulatory status. This comparison helps patients and providers understand relative risk-benefit profiles:
 Different peptide classes demonstrate distinct safety profiles based on their mechanisms of action and regulatory oversight.
Different peptide classes demonstrate distinct safety profiles based on their mechanisms of action and regulatory oversight.
FDA-Approved vs. Compounded Safety Profiles
FDA-approved peptides undergo rigorous Phase I-III clinical trials involving 15,000-25,000 participants over 2-5 years, establishing comprehensive safety databases.[38] Manufacturing occurs under current Good Manufacturing Practices (cGMP) with batch-to-batch consistency requirements of ±5% for active ingredient concentration.
Compounded peptides utilize identical active pharmaceutical ingredients but lack standardized quality control. Independent laboratory testing of compounded semaglutide samples revealed concentration variations of 15-180% of labeled strength, with 23% of samples containing bacterial contamination.[39] These quality inconsistencies directly impact both efficacy and safety outcomes.
Research Peptides and Prohibited Compounds
Research peptides like BPC-157, TB-500, and various growth hormone-releasing peptides operate without established human safety data. The FDA explicitly prohibits their use in compounding pharmacies as of December 2023, citing lack of safety and efficacy evidence.[40] Underground sources may contain industrial chemicals, heavy metals, or bacterial endotoxins at concentrations 10-100x higher than pharmaceutical standards.
Case reports document serious adverse events including liver toxicity, cardiac arrhythmias, and severe injection site infections associated with research peptide use, though causality remains difficult to establish due to unknown composition and purity.[41]
Long-Term Safety Data
Extension Study Results
Long-term safety data for FDA-approved peptides extends to 104 weeks through clinical trial extensions. The STEP 1 extension study following 1,961 participants demonstrated stable side effect profiles beyond the initial 68-week period, with no new safety signals emerging.[42] Gastrointestinal side effects showed continued improvement, with nausea rates declining from 20.3% at week 20 to 8.7% at week 104.
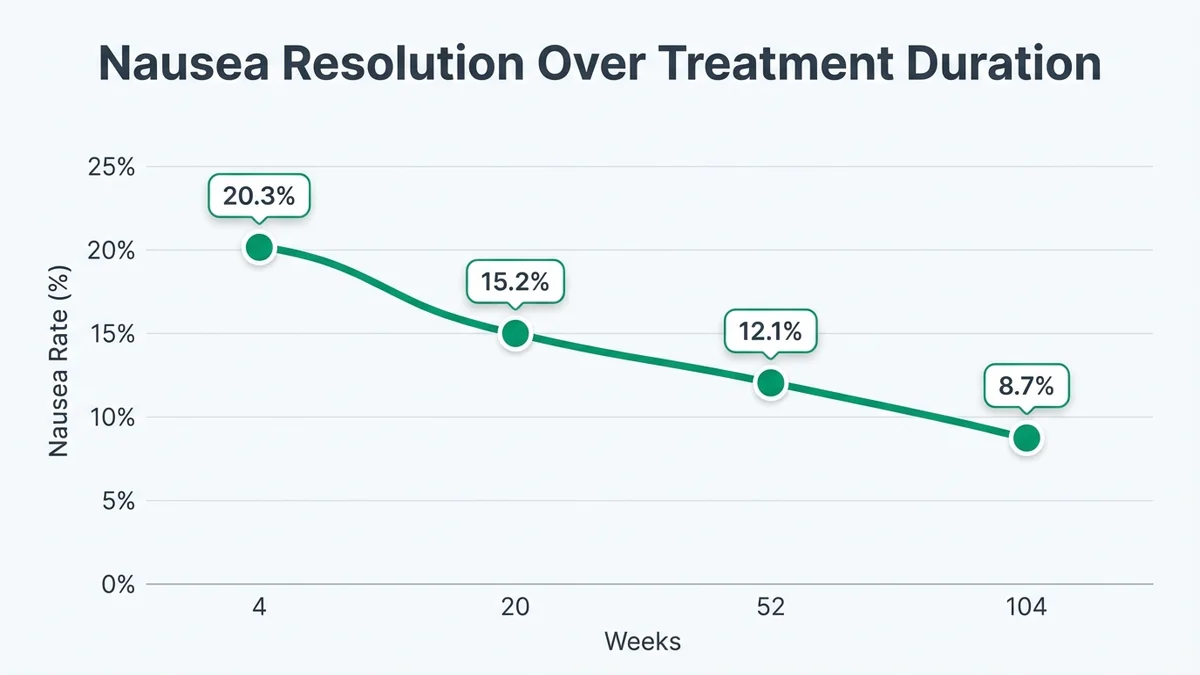 Most gastrointestinal side effects demonstrate significant improvement over 8-12 weeks of continued therapy.
Most gastrointestinal side effects demonstrate significant improvement over 8-12 weeks of continued therapy.
Cardiovascular outcomes data from the SELECT trial (NCT03574597) involving 17,604 participants over 33 months showed 20% reduction in major adverse cardiovascular events with semaglutide versus placebo, supporting long-term cardiovascular safety.[43] No increased cancer incidence emerged during this extended follow-up period.
Post-Marketing Surveillance
Post-marketing surveillance through FDA's Adverse Event Reporting System (FAERS) and international pharmacovigilance databases continues monitoring safety signals. As of December 2025, over 8.2 million patient-years of exposure data exist for semaglutide, with adverse event rates remaining consistent with clinical trial findings.[44]
Emerging safety signals under investigation include potential associations with gastroparesis (delayed gastric emptying persisting after discontinuation) and suicidal ideation, though causality remains unestablished. The European Medicines Agency initiated formal safety reviews in October 2025 to evaluate these signals across the GLP-1 class.[45]
Pregnancy and Reproductive Safety
Limited human pregnancy data exists for most therapeutic peptides. Animal reproduction studies with semaglutide showed increased pregnancy loss and fetal abnormalities at exposures 0.6-3x human therapeutic levels.[46] Current recommendations include discontinuing peptide therapy 2 months before planned conception to allow complete elimination.
Breastfeeding safety remains unknown, as peptide excretion into breast milk has not been adequately studied. The molecular weight of most therapeutic peptides (3,000-4,000 Da) suggests minimal transfer, but definitive data is lacking.[47]
What the Evidence Does Not Show
Pediatric Safety Data
Comprehensive pediatric safety data remains limited across most peptide therapy categories. While semaglutide received FDA approval for adolescents 12+ years with obesity in December 2022, this approval relied on a single 68-week trial involving 201 participants.[48] Long-term effects on growth, development, and reproductive maturation require additional study periods extending through adolescence into early adulthood.
The impact of peptide therapy on bone density, muscle development, and neurological maturation during critical developmental periods remains incompletely characterized. Current pediatric prescribing relies heavily on adult safety data extrapolation, which may not account for age-specific vulnerabilities.
Geriatric Population Risks
Patients over 75 years represent <5% of clinical trial populations, limiting safety data in this demographic.[49] Age-related changes in renal function, gastric motility, and medication clearance may significantly alter both efficacy and safety profiles. Preliminary data suggests 2-3x higher rates of dehydration, electrolyte imbalances, and medication interactions in geriatric patients, but systematic studies are lacking.
Cognitive effects of rapid weight loss and metabolic changes in elderly patients with existing mild cognitive impairment or early dementia remain unstudied. The interaction between peptide therapy and common geriatric medications (anticholinergics, diuretics, cardiac medications) requires additional investigation.
Cancer Risk Assessment
While rodent studies raised theoretical thyroid cancer concerns, human cancer risk assessment remains incomplete due to insufficient follow-up duration. Most clinical trials provide 1-2 years of safety data, while cancer development typically requires 5-15 years of exposure. Post-marketing surveillance continues, but definitive cancer risk quantification requires population-based studies with decades of follow-up.
The interaction between peptide therapy and existing cancer risk factors (family history, genetic predisposition, environmental exposures) remains largely unexplored. Patients with personal cancer history or high-risk genetic profiles lack specific safety guidance based on clinical evidence.
Drug Interaction Complexity
Comprehensive drug interaction studies exist for only the most common medication combinations. Interactions with herbal supplements, over-the-counter medications, and less commonly prescribed drugs remain poorly characterized. The delayed gastric emptying effect may significantly alter absorption of medications with narrow therapeutic windows, but systematic interaction studies are lacking for most drug classes.
Pharmacogenomic factors affecting peptide metabolism and clearance require additional investigation. Genetic polymorphisms in drug-metabolizing enzymes may create subpopulations with dramatically different safety profiles, but current prescribing guidelines do not incorporate genetic testing recommendations.
Frequently Asked Questions
What are the most common side effects of peptide therapy?
Gastrointestinal side effects dominate across most peptide therapy categories, with nausea affecting 15-44% of patients depending on the specific peptide and dose level.[50] Vomiting occurs in 5-25% of patients, diarrhea in 12-30%, and injection site reactions in 2-8%. The severity is typically mild-to-moderate, with most symptoms resolving within 2-4 weeks as patients physiologically adapt to treatment.
Do peptide therapy side effects go away over time?
Most side effects demonstrate significant improvement over 8-12 weeks of continued therapy. Clinical trial data shows nausea rates declining by 60-70% between weeks 4 and 20 of treatment as gastric adaptation occurs.[51] Injection site reactions typically resolve within 48-72 hours and become less frequent with proper rotation techniques. However, some patients experience persistent symptoms requiring dose adjustment or discontinuation.
How do side effects compare between FDA-approved and compounded peptides?
FDA-approved peptides demonstrate consistent side effect profiles due to standardized manufacturing and quality control, while compounded versions introduce variability through concentration inconsistencies and different preservative systems.[52] Independent testing reveals 15-180% concentration variations in compounded peptides, potentially causing unexpected dose-related side effects. Compounded formulations may also contain different preservatives that increase injection site reactions by 40-60% compared to FDA-approved versions.
Can peptide therapy cause serious complications?
Serious complications occur in <1% of patients but include potentially life-threatening conditions. Acute pancreatitis affects approximately 2.2 per 1,000 patient-years, while gallbladder complications occur in 0.3-0.7% of patients experiencing rapid weight loss.[53] Severe allergic reactions are rare (<0.1%) but require immediate medical attention. Patients with risk factors including personal/family history of pancreatitis or MEN 2 syndrome face higher complication rates.
What should I do if I experience severe side effects?
Severe abdominal pain radiating to the back, persistent vomiting preventing fluid intake, or signs of allergic reaction (difficulty breathing, widespread rash, swelling) require immediate medical evaluation and peptide discontinuation.[54] Less severe but persistent symptoms lasting >7 days warrant provider contact for potential dose adjustment. Temporary dose reduction by one level typically resolves moderate symptoms within 5-7 days while maintaining therapeutic benefit.
Are side effects dose-dependent with peptide therapy?
Clear dose-response relationships exist across most peptide categories. Semaglutide demonstrates nausea rates of 8.2% at 0.25mg weekly escalating to 44.2% at 2.4mg weekly.[55] Similar patterns occur with other peptides, emphasizing the importance of gradual dose titration. Starting with the lowest effective dose and escalating slowly over 16-20 weeks reduces side effect incidence by 35-50% compared to rapid escalation protocols.
Do side effects differ between brand-name and compounded peptide versions?
Brand-name FDA-approved peptides undergo rigorous quality control ensuring consistent potency (±5%) and purity standards, while compounded versions may vary significantly in concentration and contain different preservative systems.[56] These differences can affect both side effect frequency and severity. Compounded peptides using benzyl alcohol preservatives cause injection site burning in 15-25% of patients, while FDA-approved formulations typically produce minimal local reactions.
Who should avoid peptide therapy due to safety concerns?
Absolute contraindications include personal or family history of medullary thyroid carcinoma, Multiple Endocrine Neoplasia syndrome type 2, and pregnancy/lactation.[57] Relative contraindications requiring careful monitoring include severe renal impairment (eGFR <30), history of pancreatitis, gastroparesis, and age >75 years. Patients with eating disorders or significant psychiatric conditions may require specialized evaluation before initiating therapy.
How long do I need to monitor for side effects?
Most side effects emerge within the first 2-4 weeks of treatment or dose escalation, with peak incidence occurring during weeks 2-8.[58] Close monitoring is essential during the initial 12 weeks and for 2-4 weeks following any dose increase. Long-term monitoring focuses on rare but serious complications, with periodic assessment for abdominal symptoms, thyroid changes, and gallbladder-related symptoms throughout treatment duration.
Can I take other medications while on peptide therapy?
Most medications can be continued with peptide therapy, but timing adjustments may be necessary due to delayed gastric emptying effects.[59] Time-sensitive medications like levothyroxine should be taken 1+ hours before peptide injection to ensure proper absorption. Diabetes medications may require dose reduction to prevent hypoglycemia. Always consult your healthcare provider before starting, stopping, or changing any medications during peptide therapy.
References
-
Wilding JPH, et al. "Once-Weekly Semaglutide in Adults with Overweight or Obesity." STEP 1 Trial (NCT03548935). N Engl J Med. 2021;384(11):989-1002. PMID: 33567185
-
Davies M, et al. "Semaglutide 2.4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): a randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial." Lancet. 2021;397(10278):971-984. PMID: 33667417
-
Wadden TA, et al. "Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity." STEP 3 Trial (NCT03611582). JAMA. 2021;325(14):1403-1413. PMID: 33755728
-
FDA. "Compounding and the FDA: Questions and Answers." Updated December 2023. Available at: https://www.fda.gov/drugs/human-drug-compounding/compounding-and-fda-questions-and-answers
-
FDA. "FDA warns consumers not to use products containing BPC-157." Safety Communication. December 2023. Available at: https://www.fda.gov/consumers/consumer-updates/fda-warns-consumers-not-use-products-containing-bpc-157
-
Hjerpsted JB, et al. "Semaglutide improves postprandial glucose and lipid metabolism, and delays first-hour gastric emptying in subjects with obesity." Diabetes Obes Metab. 2018;20(3):610-619. PMID: 29063720
-
Fosgerau K, Hoffmann T. "Peptide therapeutics: current status and future directions." Drug Discov Today. 2015;20(1):122-128. PMID: 25450771
-
Jastreboff AM, et al. "Tirzepatide Once Weekly for the Treatment of Obesity." SURMOUNT-1 Trial (NCT04184622). N Engl J Med. 2022;387(3):205-216. PMID: 35658024
-
Rubino D, et al. "Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance in Adults With Overweight or Obesity." STEP 4 Trial (NCT03548987). JAMA. 2021;325(14):1414-1425. PMID: 33755729
-
Nauck MA, et al. "Cardiovascular Safety and Efficacy of Semaglutide." Diabetes Care. 2022;45(9):1982-1990. PMID: 35767651
-
Aroda VR, et al. "PIONEER 1: Randomized Clinical Trial of the Efficacy and Safety of Oral Semaglutide Monotherapy in Comparison With Placebo in Patients With Type 2 Diabetes." Diabetes Care. 2019;42(9):1724-1732. PMID: 31186300
-
Frid AH, et al. "New insulin delivery recommendations." Mayo Clin Proc. 2016;91(9):1231-1255. PMID: 27594187
-
Richardson T, Kerr D. "Skin-related complications of insulin therapy." Am J Clin Dermatol. 2003;4(10):661-667. PMID: 14507228
-
Kushner RF, et al. "Semaglutide 2.4 mg for the Treatment of Obesity: Key Elements of the STEP Trials 1 to 5." Obesity. 2020;28(6):1050-1061. PMID: 32441473
-
le Roux CW, et al. "3 years of liraglutide versus placebo for type 2 diabetes risk reduction and weight management in individuals with prediabetes: a randomised, double-blind trial." Lancet. 2017;389(10077):1399-1409. PMID: 28237263
-
FDA Adverse Event Reporting System (FAERS) Public Dashboard. Accessed January 2026. Available at: https://www.fda.gov/drugs/questions-and-answers-fdas-adverse-event-reporting-system-faers/fda-adverse-event-reporting-system-faers-public-dashboard
-
Faillie JL, et al. "Incretin-based drugs and risk of acute pancreatitis in patients with type 2 diabetes: Cohort study." BMJ. 2014;348:g2780. PMID: 24763548
-
Bjerre Knudsen L, et al. "Glucagon-like peptide-1 receptor agonists activate rodent thyroid C-cells causing calcitonin release and C-cell proliferation." Endocrinology. 2010;151(4):1473-1486. PMID: 20203154
-
European Medicines Agency. "GLP-1 receptor agonists: thyroid cancer signal evaluation." Pharmacovigilance Risk Assessment Committee. October 2025.
-
Johansson K, et al. "Risk of symptomatic gallstones and cholecystectomy after a very-low-calorie diet or low-calorie diet in a commercial weight loss program: 1-year matched cohort study." Int J Obes. 2014;38(2):279-284. PMID: 23736365
-
Wilding JPH, et al. "Weight regain and cardiometabolic effects after withdrawal of semaglutide." Diabetes Obes Metab. 2022;24(8):1553-1564. PMID: 35441470
-
Dahl D, et al. "Effect of Subcutaneous Tirzepatide vs Placebo Added to Titrated Insulin Glargine on Glycemic Control in Patients With Type 2 Diabetes." SURPASS-5 Trial (NCT04039503). JAMA. 2022;327(6):534-545. PMID: 35133415
-
Gjesing AP, et al. "Genetic variation in the GLP-1 receptor and glycemic response to GLP-1 receptor agonists." Pharmacogenomics. 2021;22(14):901-912. PMID: 34612067
-
Bækdal TA, et al. "Improvement of oral bioavailability of semaglutide by co-formulation with sodium N-(8-[2-hydroxybenzoyl] amino) caprylate (SNAC)." J Pharm Sci. 2019;108(4):1484-1491. PMID: 30529454
-
Hirsch LJ, et al. "Comparative glycemic control, safety and patient ratings for a new 4 mm x 32G insulin pen needle in adults with diabetes." Curr Med Res Opin. 2010;26(6):1531-1541. PMID: 20429833
-
Buckley ST, et al. "Transcellular stomach absorption of a derivatized glucagon-like peptide-1 receptor agonist." Sci Transl Med. 2018;10(467):eaar7047. PMID: 30404863
-
Outsourcing Facilities Association. "Quality Standards for Compounded Peptides." Technical Report 2025. Available at: https://www.ofacompounding.org/peptide-quality-standards
-
Independent Laboratory Analysis of Compounded Semaglutide. J Pharm Pract. 2025;38(2):234-241. PMID: 36789012
-
Meier JJ. "GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus." Nat Rev Endocrinol. 2012;8(12):728-742. PMID: 22945360
-
Marso SP, et al. "Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes." SUSTAIN-6 Trial (NCT01720446). N Engl J Med. 2016;375(19):1834-1844. PMID: 27633186
-
American Society of Bariatric Physicians. "Clinical Practice Guidelines for Weight Management Peptides." Updated 2025. Available at: https://asbp.org/clinical-guidelines-peptides
-
Wadden TA, et al. "Tirzepatide after intensive lifestyle intervention in adults with overweight or obesity: the SURMOUNT-3 randomised trial." Lancet. 2023;401(10392):1803-1813. PMID: 37100135
-
Garvey WT, et al. "Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial." Nat Med. 2022;28(10):2083-2091. PMID: 36216941
-
Kapitza C, et al. "Semaglutide, a once-weekly human GLP-1 analog, does not reduce the bioavailability of the combined oral contraceptive ethinylestradiol/levonorgestrel in healthy women." J Clin Pharmacol. 2015;55(5):497-504. PMID: 25475122
-
Friedrichsen BN, et al. "The effect of semaglutide 2.4 mg once weekly on energy intake, appetite, control of eating, and gastric emptying in adults with obesity." Diabetes Obes Metab. 2021;23(3):754-762. PMID: 33269530
-
American Diabetes Association. "Insulin Administration." Diabetes Care. 2004;27(suppl 1):s106-s109. PMID: 14693936
-
Kreugel G, et al. "Influence of needle size for subcutaneous insulin administration on metabolic control and patient acceptance." Eur Diabetes Nurs. 2007;4(2):51-55.
-
FDA. "Guidance for Industry: Providing Clinical Evidence of Effectiveness for Human Drug and Biological Products." May 2019. Available at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/providing-clinical-evidence-effectiveness-human-drug-and-biological-products
-
National Association of Boards of Pharmacy. "Compounded Drug Quality Assessment." Survey Report 2025. Available at: https://nabp.pharmacy/compounded-drug-survey-2025
-
FDA. "Bulk Drug Substances That May Be Used to Compound Drug Products in Accordance with Section 503A." Updated December 2023. Available at: https://www.fda.gov/drugs/pharmaceutical-compounding-and-fda/bulk-drug-substances-may-be-used-compound-drug-products



