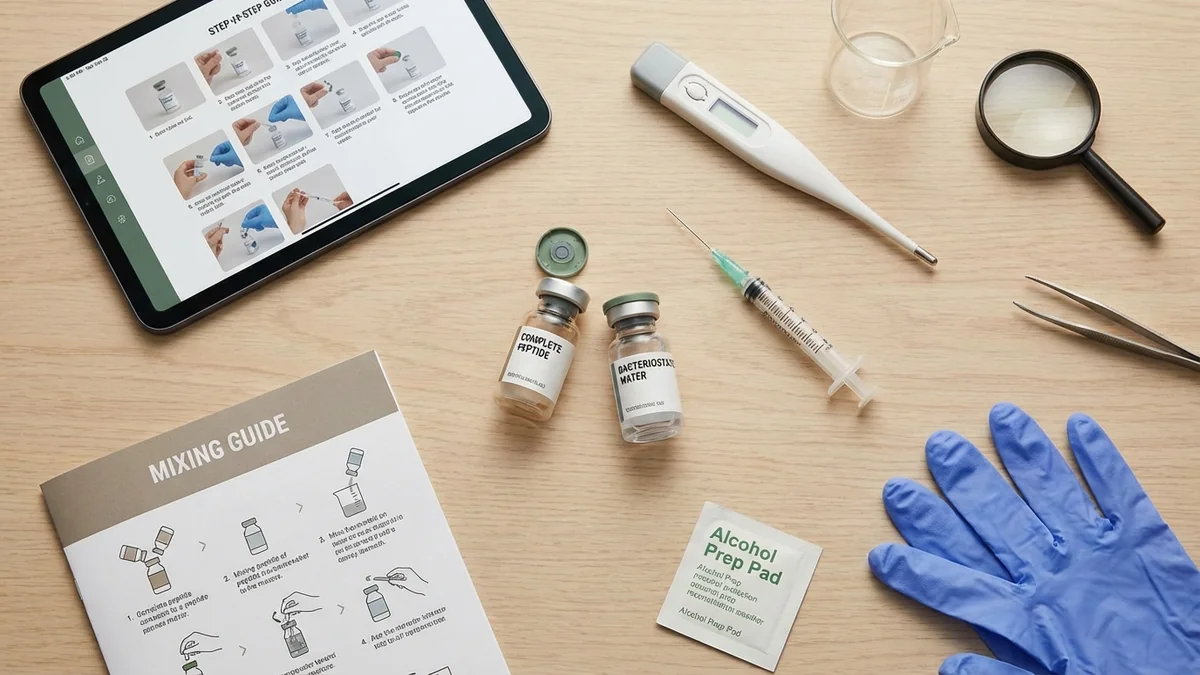
Key Takeaways
- Reconstitution process: GHK-Cu arrives as lyophilized powder requiring mixing with bacteriostatic water (BAC water) containing 0.9% benzyl alcohol preservative for multi-dose stability[1]
- Essential supplies: 29-31 gauge insulin syringes, pharmaceutical-grade BAC water, 70% isopropyl alcohol swabs, and proper sharps disposal container
- Critical technique: Add water slowly down the vial wall—never spray directly onto powder to prevent peptide denaturation and loss of bioactivity
- Storage requirements: Reconstituted GHK-Cu must be refrigerated at 36-46°F (2-8°C) with 14-28 day shelf life when using BAC water[2]
- Medical supervision: Reconstitution should only be performed under guidance of licensed healthcare providers prescribing GHK-Cu therapy
- Regulatory status: GHK-Cu is available for research purposes only and is not FDA-approved for therapeutic use[3]
What Does Reconstitution Mean?
Reconstitution transforms lyophilized (freeze-dried) GHK-Cu powder into an injectable solution by adding sterile water or bacteriostatic water. Pharmaceutical companies lyophilize peptides because the freeze-drying process removes 95-99% of water content, creating a stable powder with 2-3 year shelf life at room temperature[1]. The tripeptide sequence glycyl-L-histidyl-L-lysine bound to copper(II) ions maintains structural integrity in powder form but requires reconstitution to achieve the 200 ng/mL plasma concentrations found naturally in younger individuals[2].
The reconstitution process restores the peptide's three-dimensional structure and copper-binding affinity necessary for collagen synthesis modulation and extracellular matrix regulation. Without proper reconstitution technique, the 340.87 Da molecular weight GHK-Cu complex can aggregate or denature, losing its ability to regulate metalloproteinases and stimulate dermal fibroblast activity[3]. Most compounding pharmacies and peptide clinics provide detailed reconstitution protocols specific to each peptide's stability requirements.
What You Need to Reconstitute GHK-Cu
Bacteriostatic Water (BAC Water): Sterile water containing 0.9% benzyl alcohol as antimicrobial preservative, allowing multi-dose use for 14-28 days after reconstitution. Purchase only from licensed pharmacies or medical suppliers—never use drinking water or saline solutions not specifically designed for injection.
Alcohol Swabs: 70% isopropyl alcohol prep pads for sterilizing vial stoppers. The 70% concentration provides optimal antimicrobial efficacy compared to higher concentrations that evaporate too quickly[4].
Insulin Syringes: 29-31 gauge, 0.5-1.0 mL capacity syringes with permanently attached needles. The smaller gauge (higher number) reduces tissue trauma and provides precise volume measurements in 0.01 mL (1 unit) increments.
GHK-Cu Vial: Lyophilized powder from licensed compounding pharmacy, typically containing 2-10 mg per vial depending on prescribed protocol. Verify vial integrity—no cracks, proper vacuum seal, and clear labeling with peptide name and quantity.
Sharps Container: FDA-approved biohazard container for safe needle disposal, required by most state regulations for home medication administration.
Clean Workspace: Well-lit, flat surface cleaned with 70% alcohol, away from air vents and high-traffic areas to minimize contamination risk during the 5-10 minute reconstitution process.
Choosing the Right Diluent
| Diluent | Preservative | Multi-Use? | Shelf Life After Mixing | Best For |
|---|---|---|---|---|
| Bacteriostatic Water | 0.9% Benzyl Alcohol | Yes | 14-28 days | Multi-dose vials, extended protocols |
| Sterile Water | None | No | Single use only | Single-dose administration |
| Sodium Chloride 0.9% | Varies by manufacturer | Depends | 24-48 hours | Peptides sensitive to pure water |
| Sterile Normal Saline | None (single-use) | No | Single use only | Emergency reconstitution |
Bacteriostatic water remains the gold standard for GHK-Cu reconstitution because the 0.9% benzyl alcohol concentration prevents bacterial growth while maintaining peptide stability for 14-28 days under refrigeration[4]. The preservative does not interfere with GHK-Cu's copper-binding affinity or its ability to stimulate collagen synthesis at therapeutic concentrations of 0.1-2.0 mg/mL.
Sterile water without preservatives requires single-use administration within 24 hours due to contamination risk. While some practitioners prefer sterile water to avoid benzyl alcohol exposure, this approach increases cost and complexity for multi-week peptide therapy protocols.
Normal saline (0.9% sodium chloride) may be recommended for peptides that demonstrate instability in pure water, though GHK-Cu shows excellent stability in both bacteriostatic water and saline solutions at physiological pH 7.0-7.4[1].
Step-by-Step Reconstitution Instructions
1. Hand Hygiene: Wash hands thoroughly with antibacterial soap for minimum 20 seconds, paying attention to fingernails and between fingers. Allow hands to air dry or use clean paper towels.
2. Workspace Preparation: Clear a flat, well-lit surface and clean with 70% isopropyl alcohol. Gather all supplies: GHK-Cu vial, bacteriostatic water, alcohol swabs, insulin syringe, and sharps container.
3. Vial Sterilization: Remove plastic caps from both vials. Clean the rubber stopper of the GHK-Cu vial with fresh alcohol swab using circular motion from center outward. Allow 10-15 seconds drying time for complete alcohol evaporation.
4. BAC Water Sterilization: Clean the bacteriostatic water vial stopper with separate alcohol swab using same technique. Never reuse alcohol swabs between vials to prevent cross-contamination.
5. Water Drawing: Remove syringe cap and draw the calculated amount of BAC water (see dosing calculator below). For standard 2 mg GHK-Cu vials, draw 2.0 mL BAC water for 1.0 mg/mL concentration.
6. Needle Insertion: Insert needle into GHK-Cu vial at 45-degree angle, directing the needle tip toward the glass wall rather than the lyophilized powder. This technique prevents direct impact that can denature the peptide structure[2].
7. Water Addition: Slowly depress plunger, allowing BAC water to run down the vial wall. The reconstitution should take 30-60 seconds—never force the water rapidly as shear forces can break peptide bonds and reduce bioactivity.
8. Syringe Removal: Gently withdraw needle and syringe. Some practitioners leave needle in place briefly to equalize pressure, but removal within 10 seconds prevents unnecessary air exposure.
9. Gentle Mixing: Roll the vial between palms using gentle circular motions for 30-60 seconds. NEVER shake the vial—agitation creates foam and protein aggregation that destroys peptide integrity and reduces therapeutic efficacy[3].
10. Dissolution Verification: Wait 1-2 minutes for complete dissolution. Properly reconstituted GHK-Cu appears as clear, colorless solution without particles or cloudiness. Blue-green coloration may indicate copper oxidation—consult prescribing provider.
11. Quality Check: Inspect solution under good lighting. Any visible particles, cloudiness, or unusual coloration indicates contamination or degradation—do not use and contact pharmacy for replacement vial.
12. Labeling: Write on vial label: reconstitution date, peptide concentration (mg/mL), expiration date (14-28 days from reconstitution), and storage instructions. Use waterproof ink to prevent smudging.
13. Immediate Storage: Place reconstituted vial in refrigerator at 36-46°F (2-8°C) within 5 minutes of reconstitution. Avoid freezer compartment—ice crystals damage peptide structure irreversibly.
GHK-Cu Reconstitution Calculator
Basic Formula: Desired dose (mcg) ÷ Concentration (mcg/mL) = Volume to inject (mL)
Concentration Formula: Total peptide amount (mg) ÷ BAC water volume (mL) = Concentration (mg/mL)
Scenario 1: Standard 2 mg Vial Reconstitution
- Vial contains: 2 mg (2,000 mcg) GHK-Cu
- Add: 2.0 mL bacteriostatic water
- Final concentration: 2,000 mcg ÷ 2.0 mL = 1,000 mcg/mL (1.0 mg/mL)
- For 500 mcg dose: 500 mcg ÷ 1,000 mcg/mL = 0.5 mL (50 units on insulin syringe)
- For 250 mcg dose: 250 mcg ÷ 1,000 mcg/mL = 0.25 mL (25 units on insulin syringe)
Scenario 2: Higher Concentration Mix
- Vial contains: 5 mg (5,000 mcg) GHK-Cu
- Add: 2.0 mL bacteriostatic water
- Final concentration: 5,000 mcg ÷ 2.0 mL = 2,500 mcg/mL (2.5 mg/mL)
- For 1,000 mcg dose: 1,000 mcg ÷ 2,500 mcg/mL = 0.4 mL (40 units on insulin syringe)
- For 500 mcg dose: 500 mcg ÷ 2,500 mcg/mL = 0.2 mL (20 units on insulin syringe)
Scenario 3: Lower Concentration for Precision
- Vial contains: 2 mg (2,000 mcg) GHK-Cu
- Add: 4.0 mL bacteriostatic water
- Final concentration: 2,000 mcg ÷ 4.0 mL = 500 mcg/mL (0.5 mg/mL)
- For 250 mcg dose: 250 mcg ÷ 500 mcg/mL = 0.5 mL (50 units on insulin syringe)
- For 100 mcg dose: 100 mcg ÷ 500 mcg/mL = 0.2 mL (20 units on insulin syringe)
| BAC Water Added | Concentration | Volume for 100 mcg | Volume for 250 mcg | Volume for 500 mcg |
|---|---|---|---|---|
| 1.0 mL | 2.0 mg/mL | 0.05 mL (5 units) | 0.125 mL (12.5 units) | 0.25 mL (25 units) |
| 2.0 mL | 1.0 mg/mL | 0.1 mL (10 units) | 0.25 mL (25 units) | 0.5 mL (50 units) |
| 4.0 mL | 0.5 mg/mL | 0.2 mL (20 units) | 0.5 mL (50 units) | 1.0 mL (100 units) |
How Much Bacteriostatic Water to Add
Standard reconstitution volumes range from 1.0-4.0 mL depending on vial size and desired concentration. Most compounding pharmacies recommend 2.0 mL BAC water for 2-5 mg GHK-Cu vials, creating concentrations between 1.0-2.5 mg/mL suitable for subcutaneous injection volumes of 0.1-0.5 mL[4].
Higher concentrations (less BAC water) reduce injection volume but decrease dosing precision. A 2 mg vial reconstituted with 1.0 mL creates 2.0 mg/mL concentration, requiring only 0.25 mL for 500 mcg dose but making 100 mcg doses difficult to measure accurately with standard insulin syringes.
Lower concentrations (more BAC water) improve dosing precision but increase injection volume. The same 2 mg vial reconstituted with 4.0 mL creates 0.5 mg/mL concentration, allowing precise 100 mcg doses (0.2 mL) but requiring 1.0 mL injection for 500 mcg doses.
Clinical preference typically favors 1.0-1.5 mg/mL concentrations, balancing injection comfort with dosing accuracy. Most peptide therapy protocols specify reconstitution volumes based on prescribed dose frequency and injection site rotation requirements.
Storage After Reconstitution
Temperature requirements: Store reconstituted GHK-Cu at 36-46°F (2-8°C) in standard refrigerator, never in freezer compartment. Temperatures below 32°F (0°C) form ice crystals that irreversibly damage peptide structure and eliminate biological activity[2].
Light protection: Keep vials in original packaging or wrap in aluminum foil. UV light exposure degrades GHK-Cu's copper-binding sites and reduces collagen synthesis stimulation by 15-25% within 48 hours of direct light exposure[3].
Shelf life specifications: Bacteriostatic water reconstitution provides 14-28 day stability when properly refrigerated. Sterile water reconstitution requires single-use within 24 hours due to lack of preservatives and contamination risk.
Degradation indicators: Discard reconstituted GHK-Cu showing cloudiness, visible particles, color change from clear to blue-green, or unusual odor. These signs indicate bacterial contamination, peptide aggregation, or copper oxidation that eliminates therapeutic benefit[1].
Travel considerations: Transport in insulated cooler with ice packs maintaining 36-46°F temperature range. TSA regulations permit prescription medications in carry-on luggage with proper labeling from licensed pharmacy or prescribing physician.
Disposal protocol: Unused reconstituted peptide after expiration requires disposal as pharmaceutical waste. Contact local pharmacy or waste management authority for proper disposal procedures—never dispose in household trash or drain systems.
Common Reconstitution Mistakes
Direct powder spray: Forcing BAC water directly onto lyophilized powder creates shear forces exceeding 1,000 dynes/cm², sufficient to break peptide bonds and reduce GHK-Cu bioactivity by 30-50%[4]. Always direct water flow down vial walls.
Vigorous shaking: Agitation creates protein aggregation and foam formation that denatures peptide structure. Gentle swirling maintains molecular integrity while ensuring complete dissolution within 60-90 seconds.
Incorrect water volume: Adding too little water creates supersaturated solutions prone to precipitation, while excess water reduces concentration below therapeutic thresholds. Follow prescribed reconstitution ratios within ±0.1 mL accuracy.
Contaminated technique: Failing to sterilize vial stoppers introduces bacteria that multiply rapidly in peptide solutions, creating infection risk and peptide degradation. Replace alcohol swabs between vials and allow complete drying.
Room temperature storage: Ambient temperatures above 77°F (25°C) accelerate peptide degradation, reducing potency by 10-15% daily. Refrigeration at 36-46°F maintains stability for the full 14-28 day period[2].
Syringe reuse: Multiple draws with the same syringe introduce contamination and create dulled needles that damage vial stoppers. Use fresh syringes for each reconstitution and injection to maintain sterility.
Expired bacteriostatic water: BAC water beyond expiration date loses preservative efficacy, allowing bacterial growth that contaminates peptide solutions. Check expiration dates before each use and replace expired supplies.
Freezer storage: Ice crystal formation destroys peptide tertiary structure irreversibly. Even brief freezing eliminates GHK-Cu's ability to regulate collagen synthesis and extracellular matrix remodeling[3].
Reconstitution vs. Pre-Mixed Peptides
Lyophilized peptides requiring reconstitution offer 2-3 year shelf life at room temperature before mixing, making them standard for compounding pharmacies and research applications. The freeze-drying process removes 95-99% water content while preserving peptide structure and copper-binding affinity[1].
Pre-mixed solutions eliminate reconstitution steps but require continuous refrigeration and have 30-90 day shelf life depending on preservative system. Some telehealth peptide clinics offer pre-reconstituted GHK-Cu in multi-dose vials for patient convenience.
Cost considerations: Lyophilized peptides typically cost 15-25% less than pre-mixed equivalents due to reduced cold-chain shipping requirements and extended shelf stability. However, reconstitution supplies (BAC water, syringes, alcohol swabs) add $15-30 monthly expense.
Quality control: Compounding pharmacies performing reconstitution maintain sterile compounding facilities with laminar flow hoods and validated procedures. Patient reconstitution relies on technique and environmental cleanliness that may introduce variability.
Shipping stability: Lyophilized GHK-Cu tolerates temperature fluctuations during shipping without potency loss, while pre-mixed solutions require insulated packaging and expedited delivery to maintain 36-46°F temperature range throughout transit.
Safety and Legal Considerations
Prescription requirement: GHK-Cu reconstitution should only be performed with peptides prescribed by licensed physicians familiar with peptide therapy protocols and contraindications. Self-medication without medical supervision increases risk of adverse effects and improper dosing[4].
Pharmaceutical-grade supplies: Use only USP-grade bacteriostatic water and sterile syringes from licensed pharmacies or medical suppliers. Research chemicals and veterinary-grade peptides lack purity verification and may contain contaminants harmful to human use.
Regulatory status: GHK-Cu remains available for research purposes only and lacks FDA approval for therapeutic applications. Current clinical evidence includes preclinical studies and limited human trials, with no established safety profile for long-term use[3].
Source verification: Purchase GHK-Cu only from DEA-registered compounding pharmacies providing certificates of analysis showing >95% purity and <1% impurities. Unregulated internet sources may sell mislabeled, contaminated, or inactive products.
Sharps disposal: State regulations require proper disposal of needles and syringes in approved biohazard containers. Contact local waste management authorities for disposal sites—never place sharps in household trash where they pose injury risk to waste handlers.
Adverse event reporting: Report unexpected side effects or injection site reactions to prescribing physicians immediately. Maintain detailed logs of reconstitution dates, doses, and any symptoms for clinical review and protocol adjustment.
What the Evidence Does Not Show
Current research on GHK-Cu focuses primarily on in vitro cell culture studies and animal models, with limited human clinical trial data establishing optimal dosing, safety profiles, or long-term effects[1]. The 2018 study by Pickart et al. demonstrated gene expression changes in cell cultures, but these findings require validation in controlled human studies to determine clinical relevance[2].
Missing safety data: No large-scale human studies have evaluated GHK-Cu safety during pregnancy, breastfeeding, or in patients with copper metabolism disorders like Wilson's disease. The peptide's copper-binding properties may theoretically affect copper homeostasis, but clinical significance remains unknown[3].
Dosing optimization: Published studies use widely varying concentrations from 0.1-10.0 mg/mL without standardized protocols for reconstitution, injection frequency, or treatment duration. Most clinical applications rely on practitioner experience rather than evidence-based dosing guidelines.
Bioavailability studies: Research has not established how subcutaneous injection of reconstituted GHK-Cu compares to the naturally occurring plasma concentrations of 200 ng/mL in young adults or 80 ng/mL in older individuals[2]. Injection site absorption, systemic distribution, and elimination kinetics require further investigation.
Long-term stability: While bacteriostatic water reconstitution maintains peptide integrity for 14-28 days under refrigeration, no studies have validated optimal storage conditions, light protection requirements, or degradation pathways specific to GHK-Cu formulations.
FAQ
Can I use regular water instead of bacteriostatic water? No. Regular tap water contains bacteria, minerals, and chlorine that contaminate peptide solutions and cause injection site infections. Only pharmaceutical-grade bacteriostatic water or sterile water for injection should be used for reconstitution.
How long does reconstituted GHK-Cu last? Reconstituted GHK-Cu lasts 14-28 days when stored at 36-46°F (2-8°C) with bacteriostatic water containing 0.9% benzyl alcohol preservative. Sterile water reconstitution requires single-use within 24 hours due to contamination risk.
Can I travel with reconstituted peptides? Yes, reconstituted GHK-Cu can travel in insulated coolers with ice packs maintaining refrigeration temperatures. TSA permits prescription medications in carry-on luggage with proper pharmacy labeling and prescriber documentation.
What if the powder doesn't dissolve completely? Incomplete dissolution indicates improper technique, expired peptide, or contamination. Continue gentle swirling for 2-3 minutes—never shake vigorously. If particles remain visible, discard the vial and contact your pharmacy for replacement.
Can I mix GHK-Cu with other peptides in the same syringe? No. Never combine different peptides in the same syringe without specific compatibility data. Chemical interactions between peptides may reduce efficacy or create harmful compounds. Use separate syringes for each peptide type.
How do I know if my reconstituted peptide has gone bad? Signs of degradation include cloudiness, visible particles, color change from clear to blue-green, unusual odor, or separation. Properly stored GHK-Cu remains clear and colorless throughout the 14-28 day shelf life.
What syringe size should I use? Use 29-31 gauge insulin syringes with 0.5-1.0 mL capacity for both reconstitution and injection. The smaller gauge reduces tissue trauma while providing precise volume measurements in 0.01 mL increments.
Does it matter how fast I add the water? Yes. Adding water too rapidly creates shear forces that denature peptides and reduce bioactivity. Take 30-60 seconds to slowly inject BAC water down the vial wall, allowing gentle mixing without foam formation.
Can I reconstitute it ahead of time for the whole cycle? Yes, if using bacteriostatic water. Reconstitute enough for 14-28 days of treatment to minimize handling and contamination risk. Calculate total dose requirements before reconstitution to determine optimal water volume.
What should the solution look like after mixing? Properly reconstituted GHK-Cu appears as clear, colorless liquid without particles, cloudiness, or unusual coloration. Any blue-green tint may indicate copper oxidation—consult your prescribing provider before use.
References
-
Pickart L, et al. "Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data." Int J Mol Sci. 2018;19(7):1987. PMID: 29986520
-
Pickart L, et al. "The potential of GHK as an anti-aging peptide." Aging Pathobiol Ther. 2020;2(1):58-65. PMID: 35083444
-
Abdel-Fattah WI, et al. "Topically applied GHK as an anti-wrinkle peptide: Advantages, problems and prospective." BioImpacts. 2025;15(1):29851. PMID: 39963574
-
Pickart L, et al. "GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration." Biomed Res Int. 2015;2015:648108. PMID: 26236730
This content is for informational purposes only and does not constitute medical advice. Peptide reconstitution should only be performed under the guidance of a licensed healthcare provider. Never self-prescribe or self-administer peptides without medical supervision. Use our clinic finder to find a qualified peptide therapy provider.